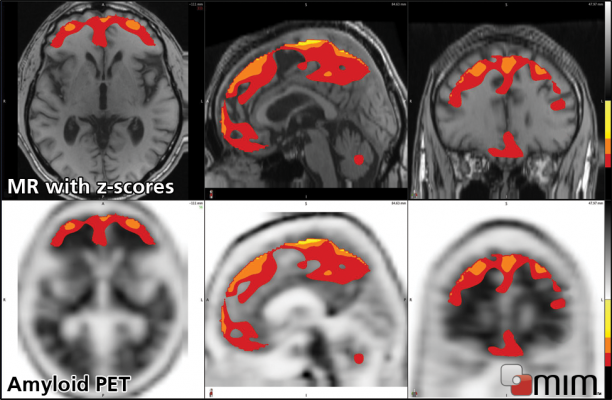
June 11, 2012 — MIM Software is currently preparing MIMneuro version 5.5.0 for release in June. MIMneuro is vendor-neutral software indicated for quantitative and statistical analysis to aid in the interpretation of PET (positron emission tomography) and SPECT (single photon emission computed tomography) brain images. The new version will include two enhancements to make it well suited for quantitative analysis of amyloid PET images.
The BrainAlign deformable registration algorithm now includes the ability to deformably register to multiple templates simultaneously. With this landmark-based deformable registration approach, local differences in anatomy are resolved more accurately than with affine registration. The variability in uptake patterns with targeted tracers such as amyloid confounds single template registration approaches. By simultaneously using multiple templates, the BrainAlign algorithm registers these images.
Additionally, MIM Software has worked with experts to define and release its third anatomical brain atlas. This probabilistic amyloid atlas is a tool specifically tuned for performing quantitative regional analysis on amyloid PET images.
MIMneuro combines these new features with existing tools – including voxel-based analysis, region-based analysis, SUVR computation, cluster analysis and surface projection analysis – into comprehensive quantitative functional neuroimaging software.
For more information: www.mimsoftware.com


 April 07, 2026
April 07, 2026 









