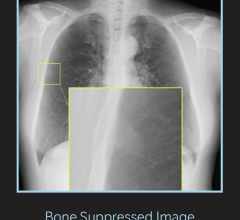
January 27, 2012 — Fujifilm Medical Systems U.S.A. introduced the FCR Prima T, the latest addition to the FCR Prima family designed to eliminate any existing barriers to the adoption of digital X-ray. Created specifically to meet the X-ray needs of low volume private practices, in the market for a compact, tabletop digital X-ray system, the Prima T comes complete with a computed radiography (CR) reader, an electronic viewing workstation and an archiving system, enabling facilities to quickly and easily transition to a softcopy imaging environments.
The system’s compact size and advanced features are the highlight and provide small practices with digital X-ray. The system is designed for small, private practice facilities looking to upgrade to digital at a low price. With dimensions measuring 22 by 21.5 by 15 inches and weighing only 86 pounds, it requires just one person to install and transport it, and can be installed on nearly any desk or tabletop.
The tabletop digital X-ray system is customized for each specialized practice to provide convenient access to the specific exams it relies on most. By offering a wider dynamic range than film, the FCR Prima T is more forgiving to over- and under- exposures so repeat exams are greatly reduced, saving both staff and patient time, and virtually eliminating unnecessary re-exposure to radiation.
There are also long-term financial and “green” benefits that facilities will immediately benefit from upon implementation. The cost of consumables is nearly eliminated, as soft-copy viewing and storage replaces the need to buy and dispose of chemicals, and there is no need to purchase and store film, allowing rooms previously devoted to file cabinets and dark rooms to now be converted into more profitable office space.
For more information: www.fujiprivatepractice.com


 March 31, 2026
March 31, 2026