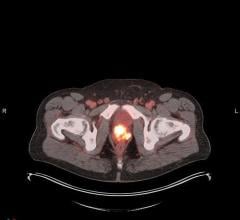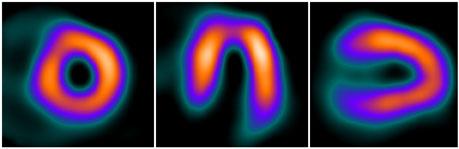
Half-dose stress images acquired with 18 mCi Tc-99 Sestamibi and 11.5 minutes scan time. Photo ourtesy of St. Lukes-Roosevelt Hospital, New York.
June 8, 2009 – UltraSPECT announced today the addition of the half-dose imaging feature to the capabilities available on the Xpress.Cardiac, its half-time WBR image reconstruction product. This new feature can minimize radiation exposure to patients, maximizing safety for both patients and staff.
With this new, FDA-approved feature, the Xpress.Cardiac now provides nuclear medicine cardiology practitioners with flexibility in both dose and acquisition time management. Depending on the patient and the physician’s preference, the rest/stress studies can be acquired using either half the radiopharmaceutical dose or half the scan time.
UltraSPECT’s half-dose capability has already been routinely utilized in a number of clinical settings in both Europe and in the U.S., with close to a 100 patients already scanned.
UltraSPECT’s Xpress.Cardiac, Xpress3.Cardiac, and Xpress/Xact.Bone products are distributed in the U.S. exclusively by Cardinal Health.
For more information: www.ultraspect.com


 June 01, 2026
June 01, 2026