March 15, 2010 - Researchers released new data from an National Institute of Health (NIH) sponsored, multi-site study of hundreds of women with newly diagnosed breast cancer shows that positron emission mammography (PEM) may reduce unnecessary breast biopsies.
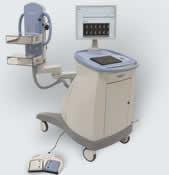
PEM scanners are high-resolution breast PET systems that can show the location as well as the metabolic phase of a lesion. This information is critical in determining whether a lesion is malignant and influences the course of treatment. Other imaging systems, such as mammography and ultrasound, show only the location, not the metabolic phase. PEM scanners, which are about the size of an ultrasound system, are manufactured by Naviscan, Inc. and have been commercially available since 2007.
The NIH-sponsored multi-site study (NIH Grant 5R44CA103102) examined women with newly-diagnosed breast cancer. Patients were accrued from six leading clinical centers across the country: ARS Johns Hopkins Green Spring, Boca Raton Community Hospital, Scripps Clinic-Scripps Green Hospital, University of North Carolina, University of Southern California Norris Cancer Center, and Anne Arundel Medical Center.
The study found that PEM was significantly more precise at identifying benign and cancerous lesions, a positive predictive value (PPV), therefore reducing the number of unnecessary biopsies. A common physician complaint regarding the use of Breast MRI is its tendency to identify suspicious lesions, requiring biopsies on lesions that ultimately are found to be benign.
This finding is a welcomed outcome for women and physicians looking for ways to reduce the patient trauma associated with biopsies and for payors looking to reduce the costs associated with unnecessary procedures. The 388 woman study showed that PEM not only demonstrated a six percent improvement in specificity at comparably high sensitivity, but that PEM also had 31 fewer unnecessary biopsies and 26 percent higher PPV than Breast MR. These results are also particularly significant for those women who cannot tolerate an MR exam and require an alternate imaging tool.
"The results of this study mean that not only do physicians have an additional, powerful tool to help treat breast cancer but that PEM is a legitimate and better alternative for the 16 percent of women who cannot tolerate MR due to claustrophobia, metallic implants, body habitus, or gadolinium reaction," said Wendie Berg, M.D., Ph.D. and Principal Investigator for the trial. Berg recently published an article in the January 2010 issue in the journal Radiology examining the reasons why high-risk women who were recommended for a MR breast screening test refused to take the exam.


 June 02, 2026
June 02, 2026 









