
Greg Freiherr has reported on developments in radiology since 1983. He runs the consulting service, The Freiherr Group.
High-performance CTs Flood Market
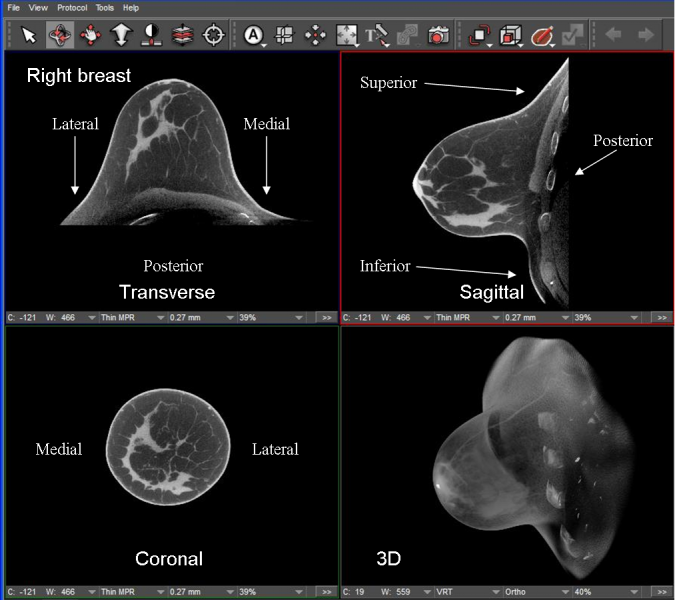
Caption: High-contrast, high-spatial resolution images of a 44-year-old woman with heterogeneously dense breast are reconstructed from a scan done with the Koning Breast CT scanner. Early this year, the FDA approved the scanner and an associated biopsy bracket. (Image courtesy Koning Corp.)
Providers under pressure to deliver low-dose, routine computed tomography (CT) can pick from the best deals ever.
Want a spectral or dual-energy CT system? You’ve got several choices. Looking for a fast trauma scanner that can do routine cardiac and neuro work? You can pick up a 64 or 128 slicer at mid-tier prices. Specialty systems are popping up, as well. One dedicated to breast imaging, approved for sale in the U.S., will be on display at this year’s Radiological Society of North America (RSNA) conference. Another is in the works for orthopedics. They’re joining the ranks of portable systems, which have been around for awhile, but recently came under new ownership.
This may be good news for buyers, at least in the short term. But industry analyst Michael Silver believes major vendors’ portfolios are bloated. “I think there are too many CT offerings,” said Silver, a senior fellow at SG2, a consulting firm that advises hospitals on equipment purchases and services.
Silver blames the global market for the glut. “Some vendors say we need one kind of offering for the Chinese market; another one for the Japanese; this for the Indian; one for the European; multiple ones for the U.S.,” he said.
Selling and supporting multiple platforms adds cost, according to Silver. “Ultimately that plays against the competitiveness of vendors in the long-term,” he said.
Meanwhile the ranks of CT vendors are expanding. Several new entrants may be at McCormick Place in Chicago in a couple months. One sure to be there is the Koning Breast CT, which the FDA approved for sale in the U.S. after extensive clinical testing as part of the Administration’s PMA (Premarket Approval) process. In announcing the approval, Koning Corporation highlighted the unique product’s ability to generate 3-D images of the entire breast without compression.
The 3-D images promise diagnostic advantages similar to those of tomosynthesis, while the lack of compression is sure to appeal to women who have long complained about discomfort during conventional mammography, which routinely uses a paddle to flatten the breast.
Not yet ready for prime time is a cone beam CT scanner that Carestream Health is grooming for orthopedic and sports medicine specialists in hospitals and clinics. A conceptual scale model of the scanner appeared in September at the British Orthopaedic Association (BOA) Annual Congress. Carestream is now optimizing the system for low-dose imaging of patient extremities, particularly knees, legs and feet. Its development is part of an initiative by the company to address what it says are unmet needs in orthopedics.
Sure to appear at this year’s RSNA are mobile CT offerings obtained by Samsung in its acquisition of Neurologica in early 2013. The 32-slice BodyTom and 8-slice CereTom are being used in emergency departments, ICUs and neurosurgical ORs, according to the company.
Value CTs from Asia are exemplified in the portfolio of Japanese vendors, such as Hitachi, Toshiba and Shimadzu, as well as Chinese vendors, notably Neusoft. Recently Chinese upstart United Imaging Healthcare (UIH) appeared on the radar of major vendors for the threat it poses to multinational brands.
The company has not registered to exhibit at this year’s RSNA. But in 2013 it opened an office in Houston, “as a first step in UIH’s global talent strategy.” Its publicly stated goal is to “recruit top medical professionals in the surrounding regions, and to promote UIH’s R&D capabilities to a new level.”
An industry intelligence report said the Chinese company’s current crop of CTs (and digital radiography (DR) systems) is as good as those of global competitors. These products and services are likely to meet with the satisfaction of “even premium customers,” according to the report.


 April 29, 2026
April 29, 2026 









