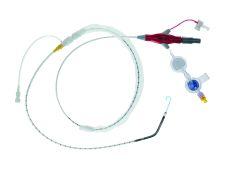
Abiomed Inc. received 510(k) clearance from the FDA for the Impella 2.5 cardiac assist device, allowing Abiomed to sell the device in the U.S.
The Impella 2.5 is cleared for use under the 510(k) for partial circulatory support for periods up to six hours. The intra-aortic balloon pump (IABP) also has 510(k) clearance and approximately 110,000 are used each year in the U.S.
The Impella 2.5 is inserted percutaneously in the catheterization laboratory via the femoral artery into the left ventricle. Up to 2.5 liters of blood per minute are delivered by the pump from the left ventricle into the ascending aorta, providing the heart with active support in critical situations.

