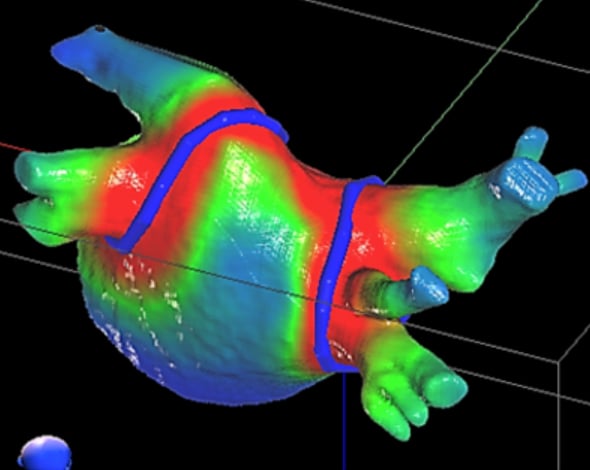
A CyberHeart cardiac ablation radiotherapy treatment plan showing where the radiation beams will ablate for a noninvasive pulmonary vein isolation procedure to treat an arrhythmia.
May 10, 2019 — Radiation oncology vendor Varian announced it acquired the start-up company CyberHeart, which has developed technology to use radiation therapy in the heart (cardiac radio-ablation) and other forms of radiosurgery for cardiovascular disease. This technology signals the company's entry into cardiology with a first of its kind technology that could represent a pardigm shift in electrophysiology (EP) ablation procedures.
The use of radiation therapy systems to pin-point target cardiac tissues to ablate would eliminate the need catheter-based ablation therapies. Which are currently the standard of care. If successful in clinical trials, this technology could make these long, involved catheter-based EP procedures obsolete, in favor of a noninvasive. Therapy where the patient does not need to be catheterized. This has been a hot topic in future innovation sessions at both the Heart Rhythm Society (HRS), American Association of Physicists in Medicine (AAPM) and the American Society for Radiation Oncology (ASTRO) the past few years.
The Cyberheart system would have application to treat atrial fibrillation, where current technologies leave room for improvement because of long procedure times of several hours and success rates only in the 70 percent range, said Hugh Calkins, M.D., FACC, FAHA, FHRS, director of cardiac arrhythmia services and professor of medicine at Johns Hopkins Hospital. He said this novel technology might be able greatly simplify and speed up Afib treatments. "It would be really amazing if it really works, but with all these things, we just need more data," Calkins said.
"Based on the early positive clinical results in ventricular tachycardia we have seen from other investigators, we believe that this technology can offer hope to cardiac arrhythmia patients," said Dee Khuntia, chief medical officer at Varian. "Cardiac radioablation would truly be a paradigm shift, bringing together two specialties—radiation oncology and cardiac electrophysiology—to collaborate in the treatment of cardiac patients."
"Varian has a long track record of innovations in the field of radiation medicine and has successfully commercialized radiosurgery technology for treating both benign and malignant lesions," said Kolleen Kennedy, president of Varian proton solutions and chief growth officer. "We look forward to expanding our focus on patient-centered innovation to the cardiac radioablation space."
First in-human studies of radioablation in the treatment of cardiac arrhythmias have been published in the New England Journal of Medicine and Circulation.[1,2]
"While cardiac radioablation technologies are not yet approved by the FDA, we feel that the results of these recent early studies are promising enough to warrant investment in this area," Kennedy said. "We're now working to evaluate the CyberHeart intellectual property portfolio and determine priorities for development and clinical trials going forward."
Related Content
New Technologies to Improve Atrial Fibrillation Ablation
New Targets for Radiation Therapy Include Cardiac AF Ablations and Renal Denervation
VIDEO: Using Radiosurgery for Atrial Fibrillation Cardiac Ablation
CyberHeart to Develop Noninvasive Cardiac Ablation Technique Using Accuray Robotic System


 May 06, 2026
May 06, 2026 









