July 29, 2014 — Balancing clinical benefit and patient-specific measurement of radiation dose received during breast screening using tomosynthesis and mammography is the focus of research presented at the 56th American Association of Physicists in Medicine (AAPM) Annual Meeting, July 20-24, 2014 in Austin. According to Volpara Solutions, the paper presents evidence that supports a workflow for breast clinics that stratifies individual woman according to their volumetric breast density (VBD) and breast size.
The AAPM research follows 19 research studies using VolparaDensity and VolparaDose that were recently presented at the 12th Intl. Workshop on Breast Imaging (IWDM 2014), including one study that reinforced the ability to generate patient-specific dose estimates using volumetric breast density for mammography and tomosynthesis.
In the study being presented at AAPM, “Which Women Based on Clinical Benefits and Dose Should Be Considered For Breast Screening with Tomosynthesis?” researchers evaluated volumetric breast density and the mean glandular dose (MGD) imparted by mammography and tomosynthesis on a patient specific basis in order to determine which women might benefit from tomosynthesis in consideration with the clinical benefits. Results demonstrated that for dense breasts, those with a Volpara Density Grade (VDG) (analogous to BIRADS) of 3 and 4, the patient-specific dose is equal or even lower for tomosynthesis than on mammography. However, for low-density breasts, the dose was significantly greater in the tomosynthesis examination than for the mammogram.
“While at higher breast density the dose is approximately equal or lower, when comparing MGD in tomosynthesis and mammography examinations at lower breast density, the mammographic dose is lower than in tomosynthesis, sometimes substantially. Since the possibility of lesion masking is directly related to breast density, clinicians might want to consider the diagnostic benefits versus the dose given in tomosynthesis,” said Ralph Highnam, Ph.D., CEO of Volpara Solutions and a co-author.
With growing emphasis on dose in medical imaging as a result of the Image Wisely initiative, it is reasonable to expect dose monitoring to expand to include mammography. The Size Specific Dose Estimation (SSDE) for computed tomography (CT) was created by an AAPM Task Group in response to the fact that reported dose is not patient-specific but the dose to a standard phantom. According to research presented at AAPM 2013, mammography units today routinely under- or over-estimate dose because they provide a dose that does not include the patient’s specific volumetric breast density in the estimation. Additionally, since the algorithms used vary, the estimations are not easily compared.
“With the breasts sensitivity to radiation, achieving diagnostically useful images while keeping X-ray mean glandular dose as low as possible is critical in breast screening. The growing adoption of breast tomosynthesis and utilization of shorter screening intervals for high-risk women has the potential to increase patients’ exposure to radiation dose. This has renewed interest in the accurate calculation and tracking of mammographic dose on an individual patient basis,” added Highnam.
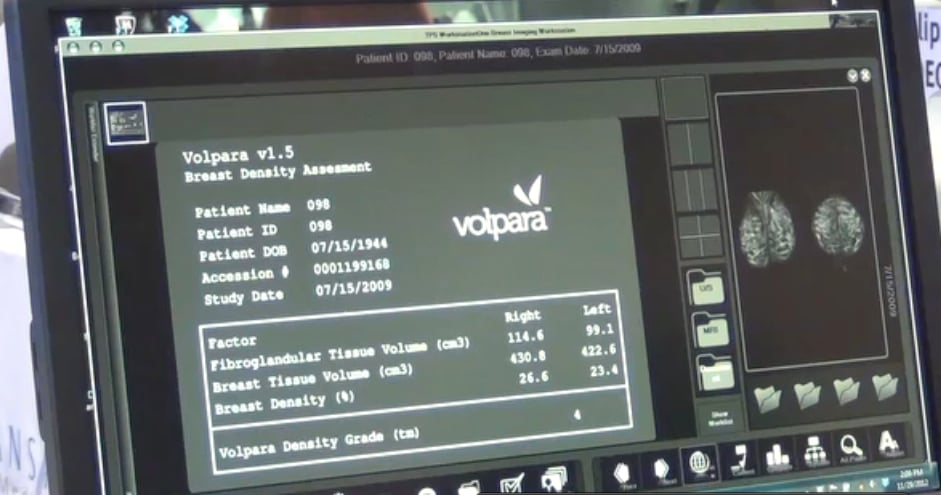
Cleared by the the U.S. Food and Drug Administration (FDA), HealthCanada, the TGA and CE-marked, VolparaDensity is used by radiologists to objectively assess density from both digital mammography and tomosynthesis images and help evaluate who might benefit from additional screening. Highly correlated to breast magnetic resonance imaging (MR) assessments, VolparaDensity is a reliable tool that automatically generates an objective, automatic measurement of volumetric breast density and a FDA cleared BI-RADS breast density category. More than 3 million women have had their volumetric breast density analyzed using VolparaDensity, which is part that enables volumetric measurements of density, patient specific dose, breast compression and other factors designed to help maintain accuracy and consistent quality in breast screening.
For more information: volparasolutions.com


 May 07, 2026
May 07, 2026 









