Cook Medical's FDA-cleared Evolution Controlled Release Esophageal Stent System is a stent designed to improve the quality of life for patients with esophageal cancer.
June 10, 2008 - Press Ganey Associates Inc. today released “The 2008 Emergency Department Pulse Report: Patient ...
June 13, 2008 - Codonics brought to SNM 2008, the Virtua Medical Disc Publisher, designed to set a new standard for ...
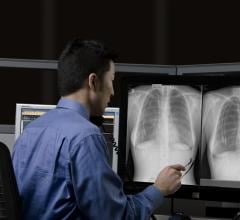
Radiology departments have many different needs and face a wide variety of challenges that can impact their departments ...
June 10, 2008 - Wright Medical Group Inc., an orthopedic medical device company, said it has acquired certain assets of ...
June 10, 2008 - The Virginia Commonwealth University Health System (VCU) recently implemented McKesson’s Horizon Medical ...
June 10, 2008 - World-wide inventors and leaders in radio frequency identification (RFID), bar coding, and other ...
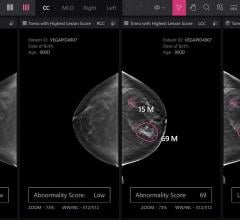
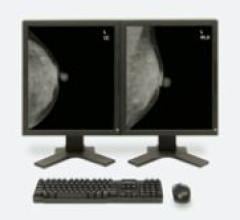
Despite decades of progress in breast imaging, one challenge continues to test even the most skilled radiologists ...
Small and large medical imaging equipment suppliers alike are struggling with slowed equipment sales resulting from ...
June 10, 2008 - The use of ultrasound contrast agents during stress echocardiograms is safe, according to results ...
iCAD’s SecondLook Digital Computer-Aided Detection system for mammography received FDA clearance for sale with FUJIFILM ...
Bayer Radiology’s Barbara Ruhland and Thom Kinst discuss how radiology departments can address the many different ...
June 10, 2008 - The U.S. Department of Health and Human Services (HHS) June 3 released two draft guidance documents for ...
June 13, 2008 - The FDA granted tentative approval for Covidien’s Abbreviated New Drug Application (ANDA) for its Kit ...
Biomarkers are key indicators for pharmaceutical companies in determining which types of drugs to develop for various ...
eHealth Saskatchewan plays a vital role in providing IT services to patients, health care providers, and partners such ...
June 10, 2008 - At the Digestive Disease Week Conference, Cook Medical announced that it has been granted 510(k) clearance from the FDA for use of the Evolution Controlled Release Esophageal Stent System, a stent designed to improve the quality of life for patients with esophageal cancer.
June 10, 2008 – The design of the University of Chicago Medical Center’s New Hospital Pavilion offers flexibility by ...
June 9, 2008 - The FDA cleared Mindray’s DC-3 color ultrasound imaging system, equipped with extensive applications in ...
The FDA cleared GE Healthcare’s new LightSpeed CT750 HD, a high-definition CT scanner that is said to produce images 100 ...
The Codonics Virtua Medical Disc Publisher is designed to set a new standard for speed, efficiency and ease of use in ...
With radiologists in short supply, the need for teleradiology services continues to grow and the market has responded in ...
June 9, 2008 - Hologic Inc. said today that it has signed a definitive agreement to acquire Third Wave Technologies Inc ...
iCAD’s SecondLook Digital Computer-Aided Detection system for mammography received FDA clearance for sale with FUJIFILM ...

 June 09, 2008
June 09, 2008