Amid the endless and monotonous droning and posturing about healthcare reform throughout this election year it’s time ...
The newly launched Back-PAK Discectomy Set is an advanced instrument system designed to make discectomy procedures and ...
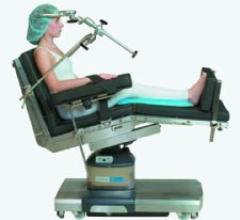
The Skytron Hercules 6701 Surgical Table provides 1,200 pounds lift and 1,000 pounds articulation and full body imaging ...
AT A GLANCE Organization: Expert Radiology Management Services, LLC Specialty: Subspecialty teleradiology — neuro and ...
Quest International Inc. recently released its QMDL5PCX for patient monitoring and information management created for ...
June 13, 2008 - MEDRAD signed a distribution and co-marketing partnership agreement with PETNET Solutions, a fully owned ...
Some healthcare facilities favor a best-of-breed approach where they assemble equipment from different manufacturers to ...
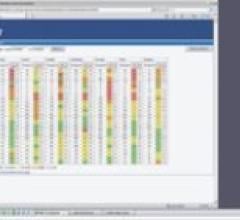
Radiology departments have many different needs and face a wide variety of challenges that can impact their departments ...
The FDA recently gave 510(k) clearance for C.R. Bard’s Agento I.C. silver-coated intermediate high volume low pressure ...
MDA TransQuest is a real-time, wireless, PDA-based patient transport dispatch system that automatically prioritizes and ...
The Canon CXDI-50G Portable DR system offers a thin, lightweight design and has a large imaging area (14-by-17 inches) ...
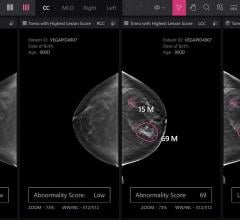
Despite decades of progress in breast imaging, one challenge continues to test even the most skilled radiologists ...
June 13, 2008 - Siemens will highlight the Symbia SPECT and SPECT/CT family of imagers, and for the first time in the U ...
June 13, 2008 – Covidien’s two new continuous positive airway pressure (CPAP) devices, the Sandman Info and the Sandman ...
Outpatient oncologists and radiation therapists may be fighting cancer in more ways and with more powerful techniques ...
Bayer Radiology’s Barbara Ruhland and Thom Kinst discuss how radiology departments can address the many different ...
Complete Care Medical Inc. released a new sterile catheter and insertion kit, which reportedly includes accessories that ...
Smiths Medical PM’s most popular medical grade quality pulse oximeter, the BCI 3301, is a portable pulse oximetry ...
ARUP Laboratories and the University of Utah’s Department of Pathology have released ARUP Consult version 2.3, a free ...
The VersaStep PLUS 15mm Bladeless Trocar is a bladeless laparoscopic access system that has a radially expandable sleeve ...
The Cmax operating table by STERIS is designed to support advancements in surgical procedures and imaging techniques ...
June 13, 2008 - Thinking Systems Corp. will showcase Thinking PACS and ThinkingRIS and SNM 2008, looking to tackle ...
June 13, 2008 - Advanced Medical Isotope Corp. (AMIC), a company engaged in the production and distribution of medical ...
As imaging modalities, such as computed tomography and magnetic resonance imaging, gain heft and horsepower that produce ...


 June 12, 2008
June 12, 2008