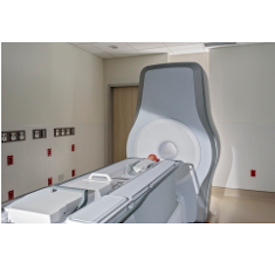
Photo: Eyas Medical Imaging
Feb. 5, 2026 — Eyas Medical Imaging, Inc. has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Ascent3T Neonatal Magnetic Resonance Imaging (MRI) System, a whole-body magnetic resonance scanner designed and optimized specifically for neonate and infant anatomy, including head, body and extremities.
“510(k) clearance marks a significant milestone for the company and reflects our mission to enable healthcare professionals to save more babies’ lives with state-of-the-art, precision imaging,” said Matt Storer, President and CEO of Eyas Medical Imaging.
The Ascent3T is the first high-field, 3 Tesla (3T) dedicated neonatal MRI system in the world. It uses a 3T magnet, enabling a more comprehensive and precise diagnostic tool that provides healthcare professionals with detailed imaging of vital anatomy, including the brain, lungs, heart, and abdomen. The Ascent3T addresses the technical limitations of using an adult-size MRI system to image babies and provides clinicians with improved ability to visualize and diagnose disease in the neonatal patient population.
A significant barrier to using MRI for neonates has been the risk of transporting fragile newborns from the neonatal intensive care unit (NICU) to alternate MRI locations within the hospital. Eyas’ innovative engineering solves that problem by delivering a whole-body 3T neonatal MRI system with a compact footprint that allows direct installation in the NICU without sacrificing image quality. Unlike most conventional MRI systems, the Ascent3T is virtually helium-free1 and does not require a quench pipe or outside venting.
The Ascent3T was conceived at one of the world’s top children’s hospitals, Cincinnati Children’s, incorporating learning from their over 1,700 infant MRI scans on prototype systems. It includes features, such as a detachable patient table that can serve as a patient transport device, providing flexibility in NICU workflow based on hospital needs and preferences. The system also leverages advanced electronics, operating software, and pulse sequences from Philips Medical Systems Nederland B.V.
“We took great care in the design of the Ascent3T. Our goal is to transform neonatal care by bringing an unprecedented level of MR imaging and access to the most vulnerable patients when and where they need it,” said MR physicist Charles Dumoulin2, Ph.D., a professor of Pediatrics and Radiology at Cincinnati Children’s and the founder of Eyas Medical Imaging.
The company is currently scaling up operations and expects to be in commercialization in the United States later in the year. The device is not yet commercially available in other countries.
- The Ascent3T requires less than 2 liters of helium.
- Dr. Dumoulin has a financial interest in the success of the Ascent3T.


 May 29, 2026
May 29, 2026 









