October 23, 2023 — Elekta announced it has acquired the Xoft business from iCAD, Inc. through an asset acquisition. By acquiring the Xoft Axxent Electronic Brachytherapy (eBx) System, together with the transfer of employees, Elekta, the world leader in brachytherapy solutions, will now be able to offer electronic brachytherapy technology and provide an expanded range of cancer treatment options.
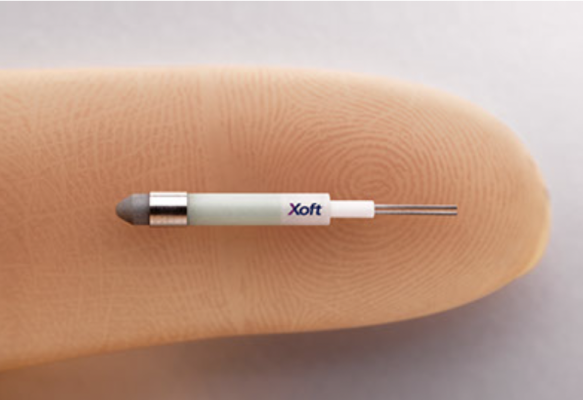
Xoft's unique technology, which uses a miniaturized low energy X-ray source instead of a radioactive isotope, can be used in a variety of clinical settings with minimal shielding. Today, it is used primarily for intraoperative treatment of breast cancer, non-melanoma skin cancer, gynecological cancers and additional clinical indications are being developed.
John Lapré, President Brachy and Neuro Solutions at Elekta, said: “Bringing Xoft’s technology into our portfolio offers exciting prospects to grow the adoption of brachytherapy and provide greater access to patients eligible for this treatment. We will be able to provide a wider range of radiation therapy options that are tailored to the specific needs and preferences of the clinics we support, while helping to reduce their operational costs – since no extensive shielding is needed – and minimize their logistical challenges such as importing radioactive material.”
The Xoft System has an installed base of more than 100 systems, compared with 1,900 Elekta Flexitron afterloaders. Elekta solutions are used in more than 120 countries and through its global network, the Xoft technology will now be able to reach more patients in need of personalized, precision radiation therapy.
The intention is to complete the transaction during the second quarter of Elekta’s fiscal year 2023/24.
The Xoft System is FDA-cleared and CE-marked for the treatment of cancer anywhere in the body using a miniaturized X-ray source to deliver a precise, concentrated dose of radiation directly to a tumor site.
For more information: www.elekta.co


 May 06, 2026
May 06, 2026 









