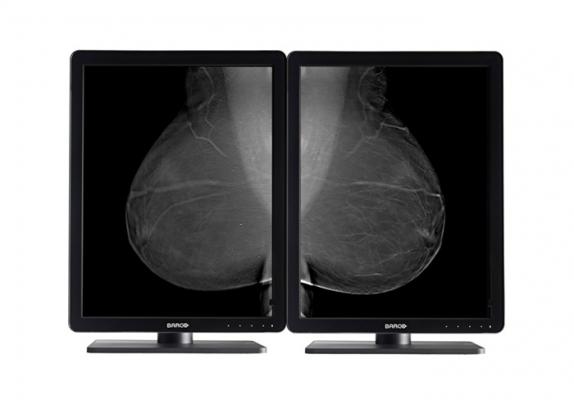
March 3, 2015 — Barco announced the launch of a new diagnostic display system, Nio 5MP LED. Cleared by the U.S. Food and Drug Administration (FDA) for radiology and mammography and featuring a number of image-enhancing technologies, the 5MP display provides excellent image quality for confident diagnoses.
Bright LED backlights help radiologists see many more shades of gray. Furthermore, Nio 5MP LED comes with a unique front-of-screen sensor, which works seamlessly with Barco’s online MediCal QAWeb solution for automated quality assurance and on-demand calibration. Additionally, Uniform Luminance Technology makes subtle details more noticeable more quickly, resulting in reduced windowing and leveling time.
Nio 5MP LED offers a high-bright LCD panel with a display resolution of 2560 x 2048 pixels and a Digital Imaging and Communications in Medicine (DICOM)-calibrated luminance of 500 cd/m² throughout its entire lifetime. It offers accurate grayscales and a high contrast ratio (1200:1) to ensure confident image reading. As part of the Nio family, the new 5MP display comes with integrated front sensor and uniform luminance technology, ensuring fast power-up and long-term luminance stability and uniformity.
On-demand image quality checks ensure maximum uptime of the display. Additionally, Barco’s LED backlights produce less heat, requiring less cooling and reducing the overall operational costs for the hospital.
For more information: www.barco.com


 May 22, 2026
May 22, 2026 









