October 31, 2011 – The Breast journal published new data from a prospective study conducted at the Seoul National University College of Medicine in Korea that shows positron emission mammography (PEM) has a higher sensitivity for tumor detection compared with whole-body positron emission tomography (PET)/ computed tomography (CT) when using both visual as well as quantitative analysis. These results were particularly striking and significant for smaller-sized tumors (less than 2 cm), likely due to the improved spatial resolution offered by PEM. This demonstrates PEM's clinical utility in being able to more effectively diagnosis early stage cancer.
According to recent journal articles, the sensitivity of PET/CT is limited, especially for low-grade and small tumors. Most PET/CT scanners currently provide a spatial resolution at 5 mm or greater, thus limiting PET/CT's ability to depict small-sized tumors such as invasive lobular carcinoma or ductal carcinoma in situ. This study provides a direct comparison within the same patient population.
A total of 113 breast lesions from 101 patients with a mean tumor size of 2.2 cm, were included in the analysis. The patients underwent PEM with FDG and whole-body PET/CT before surgical resection. The images were then analyzed visually and quantitatively using the tumor-to-normal tissue uptake ratio (TNR) with a cutoff of 2.5 or greater. The tumors were classified into 4 groups based on size using pathology measurements prior to detection sensitivity analysis for PEM and PET/CT. In visual analysis, the overall sensitivity of PEM was 95 percent, which was significantly higher than the 87 percent sensitivity found for PET/CT; ( p=0.004). In addition, the TNR of PEM was significantly higher than that of PET/CT in small tumors (<1 cm; p=0.005 and <2 cm; p<0.001).
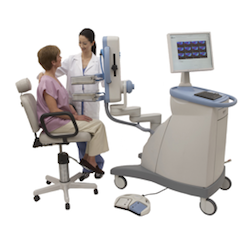
PEM is the breast application of the Naviscan high-resolution PET scanner, which both can localize as well as characterize the metabolic activity of a lesion. PEM uses parallel photon detectors in a configuration similar to mammography with breast immobilization, not compression. It is the only U.S. Food and Drug Administration (FDA)-cleared, CE Certified 3-D molecular breast imaging (MBI) device on the market. The metabolic view assists physicians in making optimal care decisions by providing an ability to distinguish between benign and malignant lesions.
For more information: www.naviscan.com


 May 08, 2026
May 08, 2026 









