Related Content
April 8, 2026 — Alpha Imaging, a Radon Medical Imaging company, will display its latest advancement in image-guided ...
February 6, 2024 — An update to the CT Colonography Reporting and Data System (C-RADS) has been published Jan. 30 in the ...
December 23, 2019 — The U.S. Food and Drug Administration cleared for marketing in the U.S. the first fully disposable ...
December 23, 2019 — Boston Scientific Corporation announced U.S. Food and Drug Administration (FDA) 510(k) clearance of ...
October 22, 2019 — OmniVision Technologies Inc. announced that its OV6948 camera module is the winner of the Guinness ...
A team of Johns Hopkins data researchers is studying the economic and safety implications associated with the devices used to perform colonoscopies as a disposable version inches closer to widespread availability.
A new study finds that trends in colonoscopy rates did not fully align with the increase in colorectal cancer (CRC) in younger adults, adding to evidence that the rise in early onset CRC is not solely a result of more detection. The study is published early online in the Journal of Medical Screening.
Check-Cap Ltd. has initiated its U.S. pilot study of the C-Scan system for prevention of colorectal cancer through detection of precancerous polyps, following Institutional Review Board (IRB) approval and full Investigational Device Exemption (IDE) application approval by the U.S. Food and Drug Administration (FDA). The first patients have ingested C-Scan, a preparation-free capsule, at the New York University School of Medicine.
Hologic Inc. announced it has received a CE mark in Europe for its Omni hysteroscope, a three-in-one modular scope with advanced visualization capabilities designed for both diagnostic and therapeutic hysteroscopic procedures. Obstetricians and gynecologists (ObGyns) can use the new Omni hysteroscope in out- and inpatient settings.
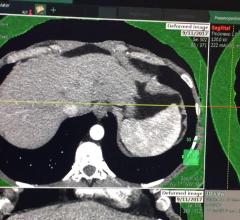
This is an example of a new endoscopic virtual peritoneal inflation tool on the patient's computed tomography (CT) ...


 April 08, 2026
April 08, 2026