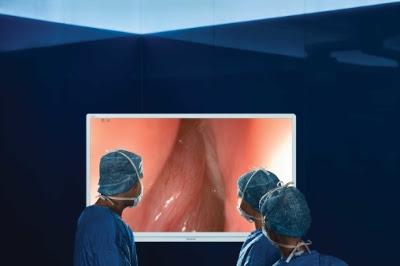
September 15, 2016 — Olympus announced that big-screen surgery with 4K UHD (ultra-high definition) endoscopy is now available for ear, nose and throat (ENT) surgeons through the Olympus Visera 4K UHD System.
Visera 4K UHD, the first of many products from the Sony Olympus Medical Solutions (SOMED) joint-venture, is a state-of-the-art big-screen surgical system that Olympus unveiled earlier this year to a broad surgical base. The system delivers four times the resolution of HD with better light and a wider color spectrum, promising to help surgeons operate with increased precision and confidence.
The only fully integrated 4K imaging chain available for healthcare use, according to the company, Visera 4K UHD allows operating room personnel to get closer to the operating field as they view cases live on a 55-inch operative display that magnifies anatomical features to deliver more visual information to the entire surgical team. It was created to address unmet needs in surgical imaging by improving the visual elements essential for sinus surgery – light, color and resolution – via an optimized 4K imaging chain that works seamlessly together to improve visibility.
Among the procedures identified as potentially benefiting from 4K UHD are Skull Base procedures (such as those for the removal of cysts or benign or malignant tumors) and Functional Endoscopic Sinus Surgery (FESS), a surgical treatment of sinusitis and nasal polyps, including bacterial, fungal recurrent, acute and chronic sinus problems. According to Truven Health Analytics Inc., there were more than 4,900,000 sinus procedures performed in the United States per year.[i] Specific to FESS, it is estimated that approximately 552,000 patients undergo the procedures annually in the United States.[ii]
For more information: www.olympusamerica.com
[i] Data for use in this 2012 study were supplied by Truven Health Analytics Inc., Ann Arbor, Mich. ("Truven Health") Any analysis, interpretation or conclusion based on these data is solely that of the authors and Truven Health disclaims responsibility for any such analysis, interpretation or conclusion.
[ii] US Market for ENT and Bronchoscopic Devices Report Suite – 2016, iData Research Inc.


 April 08, 2026
April 08, 2026