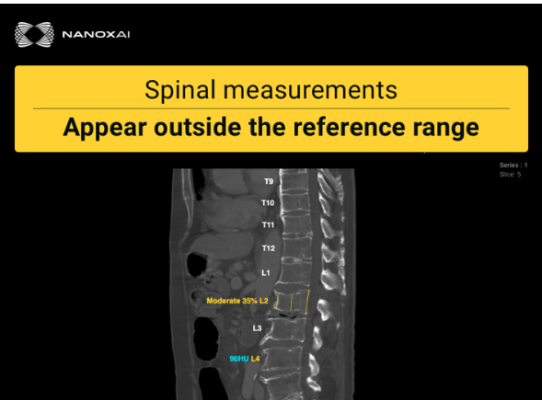
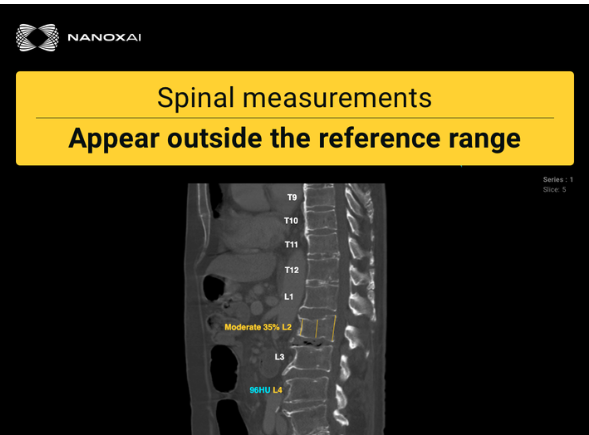
June 05, 2025 — Nano-X Imaging Ltd. has announced that its deep-learning medical imaging analytics subsidiary, Nanox AI Ltd, has received EU MDR CE (Conformité Européenne) mark certification for HealthOST, an SaMD (Software as a medical device) for bone health analysis, enabling its commercialization across Europe and ensuring compliance with the highest regulatory standards for medical software.
HealthOST is Nanox.AI’s advanced Bone Solution, which analyzes routine CT scans to assess vertebral height loss and bone mineral density. Building on its FDA 510(k) clearance received in April 2022, HealthOST leverages AI to provide qualitative and quantitative analysis of the spine from CT scans to support clinicians in the evaluation and assessment of musculoskeletal disease of the spine, such as osteoporosis, which can remain undiagnosed until life-altering fractures occur.
Since the CT scans analyzed by HealthOST were performed for other clinical indications, no additional imaging, radiation or patient time is required, making it a cost-effective screening tool. HealthOST is seamlessly integrated with existing picture archiving and communication systems (PACS), and can enable timely and appropriate preventive care.
Osteoporosis is a common progressive bone disease among adults over 50 years of age that causes bones to weaken, leading to fractures. According to the World Congress of Osteoporosis, an estimated 66% of vertebral compression fractures, a strong indicator of osteoporosis, go undetected or unreported in routine CT diagnostic reporting. Early detection is crucial, as modern treatments can improve bone strength and reduce fracture risk, helping patients maintain independence and reducing healthcare needs. This need is particularly acute in Europe, where over 25.5 million women have osteoporosis and at least 23.8 million residents are at high risk of osteoporotic fractures.i The annual cost of osteoporosis-related fractures in Europe exceeds €56 billion, with cases expected to increase 25% by 2034 due to an aging population.ii
"This CE mark for HealthOST represents a significant expansion of our AI capabilities in Europe," said Erez Meltzer, Chief Executive Officer and Acting Chairman of Nanox. "Our AI technology demonstrates the Nanox end-to-end solution that can deliver immediate practical value to opportunities for proactive care. By integrating seamlessly into routine CT scans, HealthOST helps healthcare providers maximize their existing resources, while identifying patients who might otherwise fall through the cracks of traditional screening methods."
HealthOST is the upgraded version of HealthVCF, which was evaluated in the AI-enabled Detection of OsteoPorosis for Treatment (ADOPT) study at four UK National Health Services (NHS) trusts – Cambridge, Bradford, Cardiff and Southampton – and coordinated by the long-standing clinical users at Oxford.
In the ADOPT study, HealthVCF analyzed 37,220 routine medical CT scans and identified over 3,450 new patients with vertebral compression fractures, up to six times more than the national average at NHS hospitals in the UK. These patients were asymptomatic, and the data underscores the importance of addressing the critical gap in detection of patients with vertebral fractures.
“Nanox.AI bone solution has proven to be a game-changer in bone health management," said Professor Kassim Javaid of the Nuffield Department of Orthopaedics, Rheumatology and Musculoskeletal Sciences (NDORMS), University of Oxford. "After successful integration with our clinical pathways, we saw its significant impact and chose to continue with it for another three years at Oxford. By enabling earlier identification of patients with vertebral fractures, Nanox.AI bone solution improved our system identification of patients at high fracture risk to improve patient outcomes and reduce the burden on healthcare systems through preventive care.”
Healthcare facilities interested in implementing HealthOST can visit www.nanox.vision/ai-bone-solution for more information.
i International Osteoporosis Foundation. (n.d.). Epidemiology of osteoporosis and fragility fractures. Retrieved December 13, 2024, from www.osteoporosis.foundation/facts-statistics/epidemiology-of-osteoporosis-and-fragility-fractures
ii Kanis, J. A., Cooper, C., Rizzoli, R., & Reginster, J. Y. (2021). European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Archives of Osteoporosis, 16(1), 1-10. https://doi.org/10.1007/s11657-021-00969-8


 May 21, 2026
May 21, 2026 









