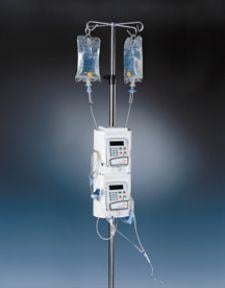
The Medrad Continuum MR Infusion System is FDA cleared for use in magnetic resonance (MR) environments up to and including 3.0 Tesla, enabling clinicians working in the latest 3.0T environments to infuse medications, sedatives and other drugs, while benefiting from the system’s ease of use and dose enhancements.
Continuum provides an expanded set of measurements so clinicians can work with a larger selection of medications, sedatives and anesthetic agents.
In addition, the infusion system introduces a new dose selection program, Non-Weight Dose, to assist in dose calculation for drugs not dependent on patient weight.
The system allows healthcare professionals to dispense cardiac stress agents at flow rates up to 1200 ml/hr and to account for time along with bolus weight and body weight with the improved bolus function.

