FujiFilm Medical Systems USA says its Fuji Computed Radiography for Mammography (FCRm) is FFDM.
The breast imaging market has been reinvigorated with an expanding arsenal of diagnostic and screening technologies for breast cancer. The recent FDA clearance of Fuji’s FCRm system, the anticipated arrival of new CR systems for mammography, not to mention GE’s newcomer — MR spectroscopy for breast — are positioned to fill the void left by FFDM and redefine breast imaging.
DMIST Sparks Trend in FFDM
Optimism in the sector has been further energized in the past seven months by the promise of full-field digital mammography (FFDM), largely due to the positive results of the Digital Mammographic Screening Trial (DMIST) conducted by American College of Radiology Imaging Network (ACRIN).
Lorna Young, MBA, senior director, Market Research, IMV Medical Information Division, speculates that the recent upswing in FFDM installations is a direct result of the ACRIN DMIST results. Market penetration of FFDM within MQSA-certified sites rose from seven percent in September 2005 to 11 percent in July 2006. “I would expect to see a similar trend with the recent clearance of Fuji’s CR mammography. One key area to watch is where CR will be installed – will it be mostly within sites that haven’t yet invested in FFDM?” asked Young.
IMV’s research also points to a continued trend toward facilities offering breast care services in an outpatient location – both hospital and nonhospital owned (freestanding) sites. IMV’s 2005 study reported that 48 percent of the surveyed sites provide mammography services in an outpatient location; of these outpatient locations, nearly 60 percent are freestanding sites. “All outpatient centers, regardless of ownership, have a lower cost of care than a hospital,” she noted.
There is also a rising occurrence in using ultrasound and MRI for breast imaging. Nearly three-quarters of the surveyed sites have ultrasound on the premises, while only 14 percent perform breast MRI procedures, either through access to fixed MRI units in their facility or using a mobile MRI service.
With the advent of CR mammography, Young believes the multimodality approach to breast imaging will certainly continue. “CR mammography is potentially a game changer,” she said, “yet we would expect to see more of an impact of this modality on breast imaging once there are at least two to three commercially available systems.” Young pointed out that FFDM didn’t really take off when only one vendor, GE Healthcare, offered FFDM, but rather there was a noticeable increase after the entry of the Hologic and Siemens FFDM systems.
CR Fills FFDM Void
Versatility and lower cost of CR-based FFDM versus existing traditional DR-based FFDM systems can meet the varying needs of mammography facilities, according to Andy Vandergrift, national marketing manager, Women’s Healthcare, FujiFilm Medical Systems USA. “Many sites have one room for mammography and cannot justify the cost of DR-based FFDM. We believe that the other available products don’t fulfill the needs of the entire marketplace, and we’ll look to fill those niches.” Vandergrift also noted that the FDA approved its Fuji Computed Radiography for Mammography (FCRm) under the digital mammography product code and that customers will be instructed to submit for reimbursement using the HCPCS codes used for digital mammography. “We want to be very clear that the FCRm is FFDM,” added Vandergrift.
The first FCRm was installed at Lenox Hill Hospital at the end of July of 2006. In mid-August of this year, Gunderson Lutheran Health System in La Crosse, WI, also installed a unit at a new clinic in Decorah, IA. According to Cathy Fischer, administrative director of the Gunderson Lutheran’s Norma J. Vinger Center for Breast Care, Imaging and Gastroenterology departments, Gunderson Lutheran, one key aspect of selecting FCRm was the lower cost compared to direct digital systems. “One FCRm unit is scalable, serving multiple mammography machines by replacing the film with the FCR cassette,” she said.
Another advantage is the ability to use the health system’s existing Fuji PACS for digital reading of images. “With most of the direct digital systems, we have to also purchase and use a dedicated workstation.” Gunderson does mammography interpretations for its main campus clinic and hospital, six regional clinics and five regional main hospitals. All FCRm images will be read at the main campus.
Vandergrift indicated that one key advantage of Fuji’s CR technology is its dual-side reading capabilities, which is important for increasing the detective quantum efficiency (DQE), resulting in less image noise and the potential for lower dose.
New CR Systems Shore Up
Although Kodak’s CR mammography systems are currently a works-in-progress in the U.S., Kodak Health Group is one of the major CR mammography players in Europe and recently launched a QA tool for CR mammography at the recent American Healthcare Radiology Administrators (AHRA) meeting in early August. Kodak will use its general radiography CR platforms for mammography and will provide an upgrade path for existing CR850, CR950 and CR975 systems, which comprise the majority of Kodak’s installed CR base.
“Our CR reader recognizes which cassette is inserted – general radiography or mammography,” said Susanne Schmitz, Kodak’s product line manager for CR mammography. Kodak will also offer a cassette that is 24-by- 30-cm to accommodate larger breasts. “All of our cassettes have rigid mounts with no bending or mechanical contact that could cause artifacts on the image.”
Konica-Minolta Medical Imaging believes it will offer end-users a broader portfolio of mammography systems with the continued development of two systems, both pending FDA clearance. The first, a CR mammography unit, has been used globally in Japan, Mexico and throughout Europe. The REGIUS CR system offers a high resolution of 43.75 micron meter and uses a higher phosphor plate than the company’s general radiography CR systems.
The second is the REGIUS PureView Mammography System, a system that may be the only medical X-ray system in the world to use a Phased Contrast Imaging technique, which enhances the edges of an image and improves contrast. By using a digital magnification technique, the REGIUS PureView Mammography overcomes the blurriness typically found in magnified images. The resulting images enable the radiologists to readily visualize the edges of the mass, calcification and fibrils. The process also increases the resolution and sharpness of the image.
“We believe this revolutionary technology will improve the radiologists’ ability to see the changes in breast tissue and increase their confidence in diagnosis,” said Eunice Lin, marketing manager for CR, Konica-Minolta Medical Imaging.
Lin also noted that with the recent change by the FDA in CR mammography’s classification from a Class 3 to a Class 2 device, it is possible that additional CR mammography products will be available for sale in the U.S. within the next year or so. Agfa also expects to introduce its CR mammography system in the U.S. market once it obtains FDA clearance.
Dedicated Breast MRI Broadens Base
Today, breast MRI is nationally reimbursed, leading to further use of the technology. One company, Aurora Imaging Technology, offers a dedicated breast MRI system. According to the company’s president, Olivia Chang, the Aurora Dedicated Breast MRI system does a better job than whole-body systems of imaging the breast because of its inherent design. Unlike a whole-body system, where the “sweet spot” exists in the center of the magnet, Aurora’s magnet was designed with a sweet spot that is oval in shape, following the natural curvature of the breast when a patient lies in a prone position.
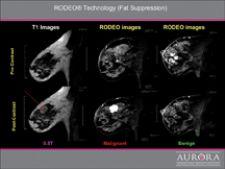
“Aurora also designed the system for uniform, simultaneous imaging of both breasts,” Chang said. The company’s RODEO technology, a fat suppression technique, lets clinicians clearly see the presence of cancer, even under the chest wall and arms where the lymph nodes are located.
For Elsie Levin, M.D., associate director of the Faulkner-Sagoff Breast Imaging and Diagnostic Centre and assistant clinical professor of Radiology, Boston University School of Medicine, accessibility, ease-of-use, patient positioning and good fat suppression are the key advantages of the Aurora Dedicated Breast MRI system. While whole-body MRI can produce quality images, Dr. Levin notes that an experienced technician for breast imaging is needed, along with the fact that breast cancer may not be a medical emergency and, therefore, the clinician may not have access to the system when desired. In her experience, Dr. Levin has found that MRI will show additional disease in 30 percent of patients preoperatively. “In half of those women, this information changes patient management,” indicated Levin.
Chang added that “the company's proprietary RODEO technology, an unsurpassed fat suppression technique, is vital for good Breast MRI and lets clinicians clearly see the presence of cancer, without the distraction of signal caused by fat.”
The Promise of MR Spectroscopy
MRI may find its greatest utility in breast imaging with the promise of MR spectroscopy. Purely a research tool until the beginning of November 2005 when GE Healthcare received FDA clearance, GE started distributing BREASE (Breast Spectroscopic Examination) as a product at the beginning of April 2006, and MR spectroscopy has since shown promising results in several clinical studies.
According to Monika Walser, MR advanced development leader for Breast Applications, GE Healthcare, BREASE is easy for the technologist to use and does not require the services of a physicist. Available on GE’s Signa HDx 1.5 T and 3.0 T systems, BREASE is currently installed at about 40 institutions worldwide, including Yale University, Edison Imaging, Atlantic Health Hospital and Seattle Radiology.
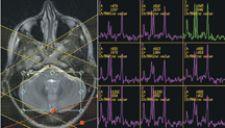
“MR spectroscopy has a potential role in breast imaging to identify the presence of choline, which is a strong indicator of malignancy. BREASE provides information to the clinician on the choline concentration in the tumor. BREASE was developed as a problem solving tool to be used in cases of equivocal contrast uptake or interminate morphology,” Walser said. A typical voxel size of 2-by-2-by-2-cm on a 1.5 T system takes approximately 5 minutes. “We also anticipate its use as an indicator in the assessment of the early chemotherapy response. However, we do need more clinical data,” and therefore GE continues to support clinical research efforts.
Additional data should be forthcoming, as Siemens Medical Solutions and Philips Medical Systems are also supporting additional research at various institutions. Siemens’ current works-in-progress MR spectroscopy is part of the company’s 1.5 T and 3.0 T Avanto, Espree and Trio systems with Tim (total imaging matrix) technology. Philips is preparing for market clearance of its works-in-progress MR spectroscopy by including the tool in the next software upgrade, Release 2, for the Achieva 1.5 T and 3.0 T MR systems. “A spectroscopy sequence will be one of the standard imaging sequences, and exam cards will be available,” said Srirama Swaminathan, Ph.D., clinical scientist, Philips Medical Systems. Release 2 will also integrate Philips’ SENSE technology for advanced post processing of the data set.
As a resident scientist at Columbia University, NY, Swaminathan is working with colleagues on multinuclei spectroscopy to further utilize the multislice acquisition of MRI to evaluate more than one region of interest or lesion. In addition to this research, he notes that the continued challenge for MR breast spectroscopy will be lipid, or fat, suppression. Swaminathan said, “Part of our works-in-progress efforts is looking at current protocols and techniques to maximize active lipid suppression.”
Siemens Medical Solution is currently testing a new works-in-progress protocol in several research sites. According to Christopher Boyea, ultra high-field marketing manager, MR Division, and Nealie Hartman, USA, MR Division, the technique – InterVIEWS – lets clinicians gather the dynamic acquisition for kinetics plus high-resolution morphologic data for DCIS evaluation in one protocol. “This will allow the clinician to gather as much information as possible about the lesion noninvasively,”
aid Boyea. He added that the company’s syngo grappa capabilities further enable clinicians to obtain high-resolution views of the breast with rapid acquisition, or “more slices at a faster rate, allowing submillimeter isotropic imaging.” Regarding faster imaging, he pointed out that researchers at NYU Cancer Center have obtained breast spectroscopy data at 3.0 T in one minute and 20 seconds and with further optimization have achieved acquiring the data in less than one minute.
Lindy Moy, M.D., radiologist and assistant professor at NYU Cancer Center, added that the center’s clinical experience with MR spectroscopy has been very positive in terms of providing another tool to evaluate enhancing breast masses seen with MRI. By enabling clinicians to measure the level of choline, Moy believes the MR can be used to successfully determine whether or not a lesion should be biopsied.
“We have enough clinical data to show that if no choline is detected, then we can avoid a biopsy,” she explained. Dr. Moy will first examine the lesion to evaluate its shape and margin and also evaluate the pattern of contrast uptake. Then, by adding the choline level data, she can further evaluate whether or not the lesion is benign. “Since this is experimental, we are conducting biopsies on each patient, but I do believe that with MR spectroscopy, we can spare unnecessary biopsies, while at the same time not miss any cancers.” Dr. Moy noted that 80 percent of biopsies are of benign tumors and that MR spectroscopy not only can lower the cost of breast cancer care to the entire healthcare system, but also reduce the mental trauma experienced by patients who have unnecessary biopsies.
FFDM Finds Future in Tomosynthesis
Tomosynthesis might hold the key for future utility of FFDM in light of breast imaging advancements with other modalities. Breast tomosynthesis is a method of performing 3-D X-ray mammography at doses similar
to conventional 2-D X-ray mammography. Tomosynthesis acquisition involves acquiring multiple images of a stationary compressed breast at different angles during a short scan. The images are then reconstructed into a 3-D series of these high-resolution slices that can be viewed individually or in a cine loop. The key promise of breast tomosynthesis is to reduce the tissue overlap and structure noise found in conventional 2-D projection mammography.
Today, all tomosynthesis systems are works-in-progress and have not been approved by the FDA for clinical use. Loren Niklason, director of tomosynthesis programs for Hologic, reports that Hologic has three breast tomosynthesis clinical sites currently in place in the U.S. with several more about to be activated. The sites are using a modified Hologic Selenia system based on a direct-to-digital selenium flat panel detector. Niklason pointed out that the clinical sites have been able to detect masses as small as three millimeters within a fatty background. “When you do see a lesion, you know exactly where it is in the breast, which will make diagnostic exams much easier.”
Dr. Steven Poplack, co-director of the breast imaging center at Dartmouth-Hitchcock Medical Center, one of the Hologic clinical sites said, “We’re looking at tomosynthesis as the next huge leap in breast imaging technology.”
With the ability to better visualize the edges and boundaries of the cancerous lesion, tomosynthesis may increase the quality of the current standard of care, says Vince Polkus, mammography marketing manager, GE Healthcare. “Tomosynthesis has strong potential to transform the practice of mammography by reducing the number of unnecessary recalls.” Polkus noted that GE is currently looking at alternative methodology to accelerate clinical evidence with technology to support widespread adoption. “We believe a strong market for tomosynthesis won’t develop unless there is a preponderance of supportive clinical evidence,” he added.
Polkus further believes the recent explosive growth of FFDM, for which ACRIN DMIST was a key catalyst, will lay the groundwork for additional research. “ACRIN provided the clinical evidence that many customers required before making the investment,” noted Polkus.
Yet, he sees greater potential beyond the 3-D acquisition technique of tomosynthesis that leads to higher specificity and sensitivity for greater lesion visualization. Polkus surmises that tomosynthesis could supplant 2-D acquisition for patient screening by unlocking the door to image fusion. “Tomosynthesis could transform breast care by allowing clinicians to fuse that information with other 3-D data, such as ultrasound, MR or optical imaging to provide more complete information for an accurate diagnosis.”
At the University of Michigan, Mark Helvie, M.D., director of Breast Imaging, is also conducting studies to further assess tomosynthesis and another works-in-progress research study on a system provided by GE Healthcare that performs full breast ultrasound simultaneously with DR-based FFDM. “While the breast is under compression, the system first takes a mammographic image, then an automated ultrasound image,” he explained. While technical issues remain by combining and correlating these two technologies, Dr. Helvie hopes that such a system would enable future diagnosis of breast cancer at the time of screening in one exam.
The growing number of techniques for breast imaging leaves it up to the clinician to determine if dedicated ultrasound, CR, MRS or tomosynthesis is the method of choice or if their combined applications are more effective in detecting breast cancer and mitigating the damage of one of deadliest diseases in women’s healthcare.


 May 01, 2026
May 01, 2026 









