According to a recent report from the Centers for Disease Control and Prevention, visits to emergency departments (ED) ...
Seca’s Model 684 Digital Multifunctional Scale facilitates safe weighing while standing, sitting or in a wheelchair ...
Mediware Information Systems has introduced its new BloodSafe system, a 510(k)-cleared, bedside point-of-care ...
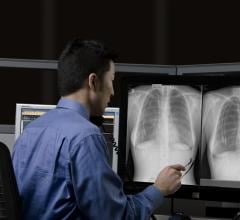
Radiology departments have many different needs and face a wide variety of challenges that can impact their departments ...
The AutoPulse is ZOLL’s revolutionary, automated chest compression system which can document, store and upload device ...
Mindray received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Hypervisor VI, a central ...
August 17, 2007 — Anesiva Inc. announced today that the FDA has approved Zingo (lidocaine hydrochloride monohydrate) ...
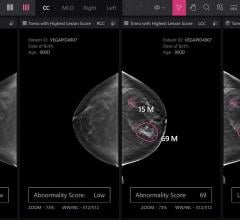
Despite decades of progress in breast imaging, one challenge continues to test even the most skilled radiologists ...
August 17, 2007 - The Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins Medicine, in MD, has placed an order to ...
The VitalCare 506NV3 series uses ComfortCuff measure-on-inflation that quickly deflates once the systolic pressure is ...
Anthro Corp.’s Zido is a compact technology cart, ready to customize for use with a PC, medical equipment or for lab ...
Bayer Radiology’s Barbara Ruhland and Thom Kinst discuss how radiology departments can address the many different ...
August 17, 2007 — Advanced BioHealing Inc. (ABH), in collaboration with leaders in wound healing research, released an ...
Baxa Corp. recently announced the expansion of its Valimed System, which uses enhanced photoemission spectroscopy to ...
Aspect Medical Systems’ FDA-cleared BIS VIEW Monitoring system is the company’s newest stand-alone monitor. The BIS VIEW ...
eHealth Saskatchewan plays a vital role in providing IT services to patients, health care providers, and partners such ...
Tensys Medical has released the latest generation of the T-Line, the TL-150Plus. The TL-150Plus provides ...
August 17, 2007 - Richard J. Rosenbluth, M. D.’s new blog site, SeniorsWithCancer.com, is dedicated to helping senior ...
August 17, 2007 – C. R. Bard Inc. announced that it has received FDA approval to market its FLAIR Endovascular Stent Graft to treat stenoses (narrowing) at the venous anastomosis of synthetic arteriovenous (AV) grafts in hemodialysis patients to increase blood flow.
Cook Medical announced today the availability of the Cook Turbo-Flo 8 French Double Lumen Central Venous Catheter ...
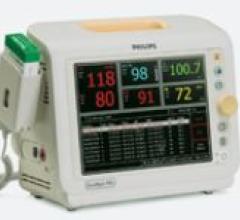
The SureSigns VS3 vital signs monitor by Philips provides caregivers with the information they require in an affordable ...
August 17, 2007 – Philips Electronics announced that it will donate an “Imagination Light Canvas,” the first of its kind ...
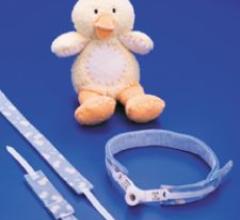
A new neonatal-infant tracheostomy tube holder that features a friendly duck pattern, reportedly easy fastener tabs and ...
August 17, 2007 — University General Hospital announced that it has been designated as a Bariatric Surgery Center of ...


 August 19, 2007
August 19, 2007