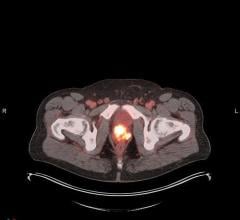
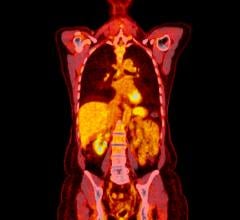
March 29, 2017 — Advanced Accelerator Applications S.A. announced that its product NETSPOT (gallium Ga-68 dotatate) has been included in the National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology version 1.2017 update for the evaluation of neuroendocrine tumors (NETs).
Eric Liu, M.D., FACS, neuroendocrine tumor surgeon and co-director, The Neuroendocrine Institute at Rocky Mountain Cancer Center and HealthOne, stated, “As a physician that sees more than 400 patients with NETs per year, I am very grateful to have NETSPOT available and included in the NCCN Guidelines. This advance in imaging capability provides treating physicians with enormous insights, enabling better directed surgeries and enhanced decision making regarding different therapeutic options. Ultimately, I believe the use of NETSPOT will lead to improved outcomes for patients.”
Lale Kostakoglu, M.D., MPH, chief, nuclear medicine and molecular imaging, at the Icahn School of Medicine at Mount Sinai, stated “We as molecular imagers are very pleased to see this valuable imaging modality be finally integrated into a national management algorithm for neuroendocrine tumors. The ability to image these patients with this compound is crucial to the success of any molecular imaging program. I believe this technology will lead to significant changes in patient management and will guide decisions for targeted therapies.”
NETSPOT, a radioactive diagnostic agent indicated for use with positron emission tomography (PET) for localization of somatostatin receptor-positive NETs, was approved by the U.S. Food and Drug Administration (FDA) on June 1, 2016, 23 months from the first pre-Investigational New Drug meeting with the agency. AAA and its radiopharmacy partners around the U.S. are now delivering 400 doses of NETSPOT per month. The company is seeking to grow its network of radiopharmacy partners from 20 sites to more than 40 sites over the first half of 2017.
In December 2016, the Centers for Medicare & Medicaid Services (CMS) granted NETSPOT Transitional Pass-Through status under an A-code (A9587) for drug reimbursement, effective Jan. 1, 2017. Additionally, the same Healthcare Common Procedure Coding System (“HCPCS”) “A Code” will be used on claims to private payers.
For more information: www.adacap.com


 May 30, 2025
May 30, 2025