May 13, 2025-- GE HealthCare recently announced the U.S. Food and Drug Administration (FDA) has approved a pediatric indication for the company’s Optison (Perflutren Protein-Type A Microspheres Injectable Suspension, USP) ultrasound enhancing agent (UEA).
Pediatric Imaging
News and new technology innovations for pediatric imaging can be found on this channel.
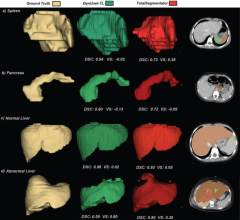
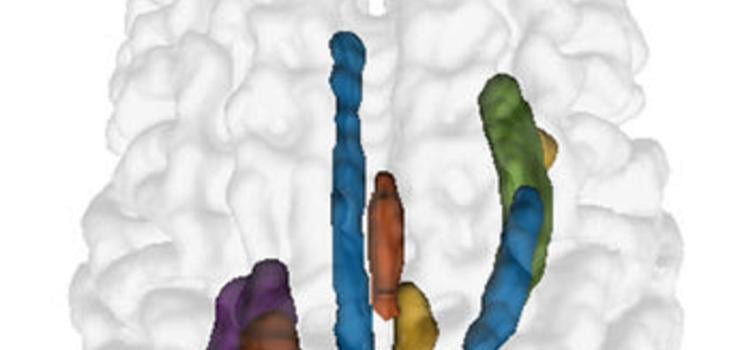
July 1, 2025 — UPDATE: The final paper is now available at: JMIR AI - ChatGPT-4–Driven Liver Ultrasound Radiomics ...
May 13, 2025-- GE HealthCare recently announced the U.S. Food and Drug Administration (FDA) has approved a pediatric ...
April 10, 2025 — Cincinnati Children’s and GE HealthCare will form a strategic research program focused on driving ...
Playing sports is good for all ages: it helps participants stay in shape, learn how to organize their time, and help ...
June 25, 2024 — Rady Children’s Hospital-San Diego, one of the nation’s top pediatric health care systems, today ...
June 24, 2024 — Well-known Chicago and worldwide philanthropist Ann Lurie, namesake of the Ann and Robert H Lurie ...
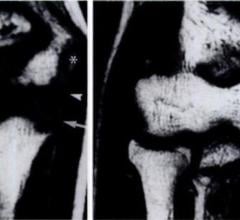
June 13, 2024 — Cervical spine injuries in children are relatively rare but can have serious consequences, like ...
Medical imaging plays an increasing role in the accurate diagnosis and treatment of numerous medical conditions. The ...
May 15, 2024 — Transfer learning (TL) models trained on heterogeneous public datasets and fine-tuned using institutional ...
May 2, 2024 — Head and abdominal trauma is a leading cause of death for children. About 1%–2% of children who come to ...
April 16, 2024 — The Society of Interventional Radiology (SIR) has published a new position statement outlining best ...
FUJIFILM Medical Systems U.S.A., Inc. and FUJIFILM SonoSite Inc., are offering a full-suite pediatric solutions ...
March 6, 2024 — Lantheus Holdings, Inc. (Lantheus), a radiopharmaceutical company, has announced that the U.S. Food and ...
January 26, 2024 — InkSpace Imaging, a leader in innovative diagnostic medical device technology, is proud to announce ...
Playing sports is good for all ages: it helps participants stay in shape, learn how to organize their time, and help ...
It looked like the autistic child at the Children's Hospital of Georgia at Augusta University (AU) Health would have to ...
December 1, 2023 — The Radiation Oncology Program at Children’s Hospital Los Angeles is one of only a few in the country ...
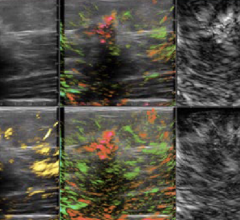
November 30, 2023 — Youth baseball players are prone to elbow pain and injuries, including repetitive overuse changes ...
November 29, 2023 — Using artificial intelligence (AI) to analyze specialized brain MRI scans of adolescents with and ...
November 21, 2023 — A newly developed artificial intelligence (AI) system that analyzes specialized MRIs of the brain ...
August 29, 2023 — NorthStar Medical Radioisotopes, LLC (“NorthStar"), a global innovator in the development, production ...
August 29, 2023 — Mammograms are an essential part of preventive healthcare, and when an initial review reveals a ...
August 23, 2023 — With the goal of ensuring access to safe and clinically useful AI for all pediatric patients, the ...
August 18, 2023 — According to an accepted manuscript published in the American Journal of Roentgenology (AJR), an ...





 July 01, 2025
July 01, 2025