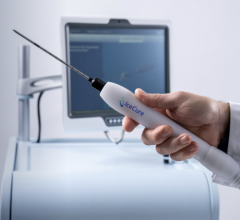
January 7, 2016 — A study published by the Journal of Medical Virology demonstrates that the trophon EPR is the only high-level ultrasound disinfection system proven to kill natural, infectious, high-risk, cancer-causing strains of human papillomavirus (HPV).
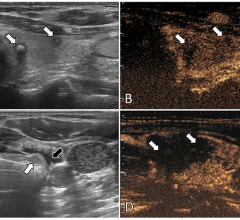
High-risk HPV accounts for 5 percent of all cancers worldwide, is responsible for almost all cases of cervical cancer and is a leading cause of oral, throat, anal and genital cancers.
A number of published studies have already demonstrated there is a real risk of cross contamination between patients examined with ultrasound probes. The studies showed up to 7 percent of vaginal ultrasound probes remained contaminated with HPV despite the use of probe covers and routine disinfection. HPV is also a very stable virus and is able to remain infectious on fomites and surfaces for days despite routine disinfection.
Testing of natural, infectious “real” HPV has not previously been possible due to the difficulties of growing enough of the natural, infectious virus for research.
This situation changed recently when researchers from Penn State College of Medicine and Brigham Young University in the United States developed a method to grow HPV. “We were the first ever to grow papillomavirus in culture,” said Craig Meyers, distinguished professor of microbiology and immunology, Penn State College of Medicine. “In our original study we chose, specifically, glutaraldehyde and ortho-phthalaldehyde (OPA) to test because these are disinfectants that hospitals believe are sterilants that kill everything, including papillomavirus.”
This original study, which was published in 2014, showed both glutaraldehyde and OPA completely failed to inactivate natural, infectious, high-risk HPV16 – even after 24 hours of contact time. The most recent study used a stringent method in line with U.S. Food and Drug Administration (FDA) requirements for virucidal testing. OPA was retested and the trophon EPR device tested against HPV16 and HPV18, which are the major cancer-causing types of HPV. The results showed that the trophon EPR was the only disinfectant to completely inactivate HPV.
“Our first study showed that commonly used disinfectants are not effective, while this latest study found that trophon EPR was completely effective. The concern is that these other liquid chemical disinfectants are commonly used in medical and healthcare facilities. Where this is happening, HPV is not being killed, posing a risk for transmission,” said Meyers.
The study concluded that healthcare professionals should carefully consider these results when selecting appropriate disinfection methods and that a review of disinfection standards with respect to high-level disinfection may be warranted.
“Publication of the study is very timely given the current increased scrutiny of disinfection practices, said Michael Kavanagh, Nanosonice CEO and president. “An alert issued in the U.S. by the Centers for Disease Control and the FDA in September stipulates that reusable medical devices, such as ultrasound probes, must be properly maintained, cleaned and disinfected/sterilized and urged healthcare facilities to immediately review current reprocessing practices to protect patients.”
According to Kavanagh, the trophon technology uses a proprietary disinfectant containing a high concentration of hydrogen peroxide that is sonically activated to form a sub-micron particle mist. The mist generates a high number of free radicals with superoxidative properties that destroy the virus. Traditionally, ultrasound probes are soaked in toxic liquid chemicals or manually wiped for a few minutes with a pre-impregnated wipe.
For more information: www.nanosonics.com.au

 April 24, 2024
April 24, 2024