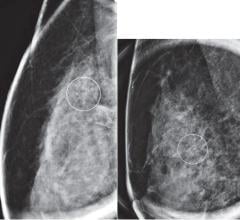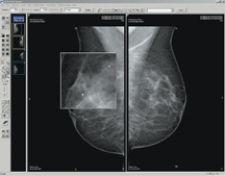
eRAD has received FDA 510(k) clearance for soft-copy viewing of digital mammography images. This clearance allows eRAD to market its eRAD PACS Diagnostic Workstation software and FDA-cleared display hardware to the mammography community. In conjunction with this clearance, eRAD is adding several features to its viewing capabilities, including mammography-specific hanging protocols, mirrored rendering tools including panning and magic glass zooming, support for R2 Technology’s ImageChecker CAD markers and BI-RAD support in reports. The product enhancements provide efficient image review, distribution and storage for full-field digital mammography (FFDM) systems.


 May 10, 2024
May 10, 2024