If you enjoy this content, please share it with a colleague
RELATED CONTENT
News | Ultrasound Imaging
October 9, 2017 — A clinical trial conducted at Stanford University Medical Center published online in Anesthesia & ...
Technology | Radiology Imaging
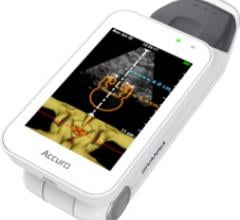
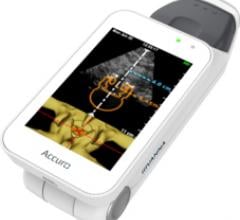
Rivanna Medical announced that it has received U.S. Food and Drug Administration (FDA) 510(k) clearance to market Accuro, a handheld and untethered smart phone-sized ultrasound device. Accuro is designed to guide spinal anesthesia with automated 3-D navigation technology in addition to ultrasound imaging of abdominal, musculoskeletal, cardiac and peripheral vascular anatomies.

 October 09, 2017
October 09, 2017