If you enjoy this content, please share it with a colleague
InfraScan Inc.
RELATED CONTENT
InfraScan, a medical device firm specializing in brain injury diagnostic products, signs a $3.7 million, 4-year contract with the U.S. Marine Corps and the Navy. The project provides for advanced technology development, and field evaluation activities to develop a next-generation version of the company's Infrascanner brain hematoma detector.
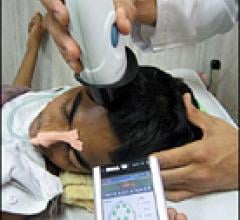
December 14, 2011 – The U.S. Food and Drug Administration (FDA) allowed marketing of the first hand-held device intended to aid in the detection of life-threatening bleeding in the skull called intracranial hematomas, using near-infrared spectroscopy. The device, the Infrascanner Model 1000, can help healthcare providers identify patients with critical head injuries who need an immediate computed tomography (CT) brain imaging study.
July 20, 2010 – InfraScan announced this week that it has signed a $2 million contract with the U.S. Marine Corps and Navy to develop a next-generation version of the company’s Infrascanner brain hematoma detector.


 October 20, 2014
October 20, 2014