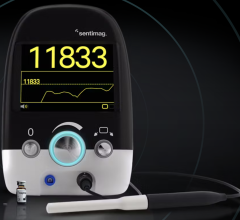
November 20, 2017 — Eizo Corp. announced it has received U.S. Food and Drug Administration (FDA) 510(k) clearance for breast tomosynthesis and digital mammography for its 5-megapixel color medical monitor, the RadiForce RX560.
Watch the VIDEO Eizo Introduces the RadiForce RX560 at RSNA 2017.
Breast cancer screenings increasingly combine the use of mammography and ultrasound to view patients with high breast density. Furthermore, if breast cancer is suspected, various other tests may also be needed such as biopsy, breast magnetic resonance imaging (MRI) and computed tomography (CT). Because of this, monitors that can accurately display tomosynthesis, mammography and general radiology are becoming increasingly important.
The RX560 is the world's first medical monitor to use a low temperature polysilicon (LTPS) liquid crystal display, according to Eizo. Because of this, the color monitor can reach a brightness of up to 1100 cd/m2, similar to that of monochrome monitors. In order to detect the smallest structures within breast tissue, the monitor offers a high contrast ratio of 1,500:1. Furthermore the monitor has a response time of 12 milliseconds, making it ideal for enlarging and panning medical images, as well as quickly displaying 3-D breast tomosynthesis which is comprised of several images in each shot. The 5 megapixel (2,048 x 2,560) resolution is ideal for viewing the fine details in breast images.
The RX560 is able to display both monochrome and color medical images on the same screen due to the Hybrid Gamma PXL function included with the monitor. With this function, both monochrome and color images are set to the optimum grayscale and gamma curve pixel-by-pixel, improving the efficiency of interpretation work.
Eizo will demonstrate the RadiForce RX560 at the 103rd Radiological Society of North America Annual Meeting (RSNA 2017), Nov. 26-Dec. 1 in Chicago.
For more information: www.eizo.com

 May 10, 2024
May 10, 2024