March 6, 2019 – With the passage of HB1124, South Dakota becomes the 37th state with breast density inform legislation. The law, which was signed on March 5, 2019 by Gov. Kristi Noem, goes into effect July 1, 2019.
South Dakota Rep. Taffy Howard introduced the legislation after becoming alarmed about the inconsistency of density notification across the state and the lack of information about the risks associated with dense breasts.
“Breast density is a critical piece of information regarding a woman’s breast health, but women are reliant upon their healthcare provider for this information, which is why this legislation was so vital. Many women across this state are informed by their provider about their breast density, but not all. This bill ensures all women across South Dakota are informed as to their breast density. It will also provide additional information if they are in one of the two higher levels of density and encourage women to discuss findings with their primary care providers,” said Howard.
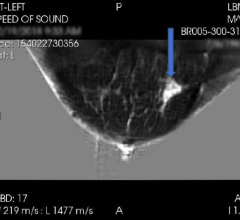
Approximately 40 percent of women aged 40 and over have dense breast tissue that may hide cancers on mammography. Women with dense breasts may benefit from supplemental screening tests, in addition to mammography, to detect cancers. Women with extremely dense breasts are also 4-6 times more likely to develop breast cancer than women with the least dense breasts. Providing women with their breast density classification would enable them to initiate discussions with their physicians about whether supplemental screening might be beneficial.
“Sen. Deb Soholt was tremendously helpful in getting key players on board,” continued Howard. “I am tremendously grateful to JoAnn Pushkin, executive director of DenseBreast-info.org for all her hard work in calling attention to this issue and, along with Jill Schultz, with Avera Breast Center, for providing outstanding testimony in support of the bill. We all have an obligation to fight this disease in whatever way we are able, and I was glad to be able to aid in a small way in that fight.”
South Dakota’s state law follows the Feb. 15, 2019 passage of federal legislation directing the U.S. Food and Drug Administration (FDA), through the regulatory process, to develop reporting language and ensure that mammography reports and summaries received by patients and their providers include appropriate information about breast density. The timeline for implementation of federally mandated reporting language is unknown; however, once the new standard is introduced, individual state law review will be necessary.
Read the article "Congress Directs FDA to Establish Federal Breast Density Inform Standard"
For more information: www.densebreast-info.org


 April 18, 2024
April 18, 2024