December 20, 2018 — Seno Medical Instruments Inc. (Seno Medical) reported results of a study demonstrating that morphologic and functional opto-acoustic (OA/US) features provide a non-invasive approach to helping distinguish breast cancer molecular subtypes. The data were presented at the Radiological Society of North America (RSNA) 2018 annual meeting, Nov. 25-30 in Chicago, and the abstract was featured as one of three High Impact Clinical Trials selected for this special session.
There are four distinct main molecular subtypes of breast cancer, which are defined based on the presence of hormone receptors (estrogen [ER] and progesterone [PR]) with or without human epidermal growth factor receptor 2 (HER2) protein overexpression or extra copies of the HER2 gene, and elevation of the ki67 proliferation marker. The four subtypes are:
-
Luminal A (LumA; ER+/HER2−negative, ki67<14%);
-
Luminal B (LumB; ER+/HER2-negative, ki67≥14%, or ER+/HER2+);
-
HER-2 amplified or positive (HER2+; ER-negative, HER2+); and
-
Triple negative breast cancer (TNBC); ER and PR-negative/HER2−negative).
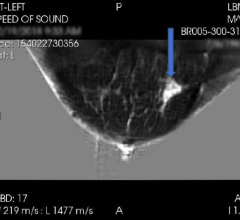
The study investigated the potential role of functional OA/US imaging-derived hemoglobin de-oxygenation and angiogenesis feature scoring combined with conventional gray-scale ultrasound (US) in non-invasively diagnosing these breast subtypes.1 A total of 2,105 women with suspicious breast masses who underwent pre-biopsy OA/US imaging using Seno's Imagio OA/US Breast Imaging System at 16 clinical sites between December 2012 and September 2015 were included in the study. Lesions revealing invasive breast cancer on needle biopsy were retrospectively reviewed, and seven blind readers scored the internal (OAINT) and external (OAEXT) OA/US features of identified cancers. The ratio of total internal to total external US and OA/US feature scores (RInt/Ext) was derived. Tumor hormone receptor (ER and PR), and HER2 status, and available ki67(%) labeling index were derived from pathology specimens. Key findings from the study include:
-
Of 653 invasive cancers, 537 (82.2 percent) were ER+ and 111 (17 percent) were ER-negative, with ER data missing for 5 (0.8 percent) patients;
-
ER+ cancers had significantly higher OAEXT (p<0.001), with lower OAINT (p<0.05) and RInt/Ext (p<0.001) compared with ER-negative ones;
-
Of 532 patients with available pathologic molecular subtype, 186 (35 percent) were LumA, 244 (45.9 percent) were LumB, 79 (14.8 percent) were TNBC and 23 (4.3 percent) were HER2+;
-
All OAEXT scores were lower in TNBC compared with LumA (p<0.001), whereas OAINT vessels and total scores were lower in LumA compared with TNBC (p<0.05);
-
The mean RInt/Ext was significantly higher in TNBC (1.7, SD ± 0.7) compared with LumB (1.3, SD ±0.5) and LumA (1.2, SD ± 0.5) subtypes (p<0.001), but not significantly different from HER2 (1.5, SD ±0.6); and
-
RInt/Ext feature scores helped distinguish LumA vs. LumB (p<0.05), LumA vs. HER2+ (p<0.05, LumA vs. TNBC (p<0.001) and LumB vs. TNBC (p<0.001.
The data demonstrate that morphologic and functional OA/US features may provide a non-invasive approach to help distinguish breast cancer molecular subtypes. The study authors conclude that data from such subtype analyses could potentially facilitate clinical management decisions.
"The exciting finding in this study is OA/US imaging features may serve as imaging biomarkers that predict molecular phenotypes. This not only helps establish the diagnosis of breast cancer, but also distinguishes cancers with poor prognosis from those with good prognosis non-invasively – using no radiation, contrast or a need for biopsy," said Basak E. Dogan, M.D., University of Texas Southwestern Medical Center, presenter at RSNA and the first author of the study. "This is an important step forward to non-invasively optimize treatment regimens in keeping with the unique molecular profile of each woman's cancer. Furthermore, it is a great opportunity to assess the cancer three dimensionally instead of making treatment decisions based on small biopsy samples. OA/US has the potential to improve treatment outcomes for women with invasive breast cancer."
The Imagio OA/US Breast Imaging System is the subject of a U.S. premarket approval (PMA) filing with the U.S. Food and Drug Administration (FDA) and does have European CE Mark.
For more information: www.senomedical.com


 April 18, 2024
April 18, 2024