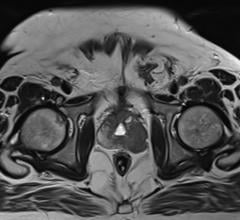
February 21, 2019 — ViewRay Inc. received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market new soft tissue visualization capabilities for its MRIdian image-guided radiation therapy (IGRT) system.
The new upgradable capabilities enhance the features of MRIdian's SmartVISION MRI (magnetic resonance imaging), including:
-
Expanded high-definition visualization and enhanced contrast between different tissues, to assist clinicians with tissue visualization and beam contouring;
-
The potential to aid in the assessment and prediction of tumor response to radiation therapy is enabled by "DWI", a diffusion weighted imaging feature that tracks treatment progress by distinguishing between tumor and normal tissues;
-
Faster, brighter, more detailed anatomical planar imaging to strike tumors with greater precision and accuracy through proprietary technology, which allows for a 2X increase in MRI speed (to 8 frames per second), a 2X increase in image resolution, and a 2X improvement in MR signal-to-noise ratio (SNR); and
-
Potential reduction in treatment delivery time through enhanced multileaf collimator (MLC) speed.
MRIdian's SmartVISION provides high-definition, diagnostic-quality MR imaging. SmartVISION was designed to maintain high-fidelity beam delivery while mitigating the risks of skin toxicities, as well as trapped or distorted dose. MRIdian's SmartADAPT helps allow clinicians to generate daily MR setup scans in seconds and leverage high-contrast anatomical detail to rapidly reshape dose delivery based on the current position of both the tumor and adjacent critical structures – all while the patient is in the treatment position. MRIdian's SmartTARGET visualizes the tumor's edges and surrounding organ position in real-time using a non-ionizing, streaming video perspective. When tumors or organs-at-risk change shape or position, SmartTARGET instantly reacts, automatically controlling beam delivery.
For more information: www.viewray.com
Related MRIdian Content
VIDEO: Clinical Use of the ViewRay MRIdian Linac System at Henry Ford
VIDEO: MRI-Guided Radiation Therapy Trial for Pancreatic Cancer


 April 04, 2024
April 04, 2024