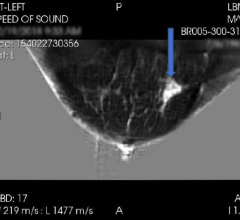
February 19, 2019 — As part of a federal spending bill passed late Friday, Congress directed the U.S. Food and Drug Administration (FDA) to establish a national minimum standard for including fibroglandular breast density information on mammography reports. While 36 states have already enacted their own breast density inform legislation, the federal version will ensure that every state takes a minimum level of action on making patients aware of the increased breast cancer risk associated with dense breasts.
The law directs the FDA, through the regulatory process, to develop language for reports and summaries from mammography facilities to include with appropriate information about breast density for patients and their providers. According to DenseBreast-info.org, the information must include, at a minimum:
-
The effect of breast density in masking the presence of breast cancer on a mammogram;
-
The qualitative assessment of the patient’s fibroglandular breast density by the provider who interprets the mammogram according to the American College of Radiology Breast Imaging-Reporting and Data System (BI-RADS); and
-
A reminder to patients that individuals with dense breast tissue should talk with their providers if they have any questions or concerns about their summary.
“As part of the funding bill Congress passed yesterday, the FDA must now ensure mammography reports include appropriate breast density information. Dense tissue can hide cancer on mammograms, so this information is vital to catching breast cancer early,” said Sen. Dianne Feinstein (D-Calif)., a longtime supporter of the breast density inform movement who championed the inclusion of the density standard in the current spending bill in the Senate. Rep. Rosa DeLauro (D-Conn.) led efforts in the House.
The grassroots movement to spread information about breast density began in 2009 when Connecticut became the first state to enact a breast density inform law. Those efforts were spearheaded by Nancy Cappello, Ph.D., who was diagnosed with stage 3C breast cancer in February 2004, just six weeks after her annual mammogram came up normal. Her doctor told her the cancer was missed on the mammogram because she had dense fibroglandular breast tissue, a concept that was little-known among patients at the time. Cappello passed away in November from secondary myelodyspastic syndrome (MDS), a bone marrow cancer that was a complication of her prior aggressive breast cancer treatments.
Watch the VIDEO: The Impact of Breast Density Technology and Legislation, an interview with Cappello at RSNA 2017.
Today, 10 years later, the movement has expanded to 36 states, covering an estimated 89 percent of women in the U.S. Each state law has slightly different requirements, however, for how much information is given to patients and providers. According to JoAnn Pushkin, executive director of DenseBreast-info.org, some laws only require that women are informed about breast density generally and not their own density and associated cancer risk.
“Implementation of this national law will help to address this disparity, as all imaging facilities that perform mammography screening will be required to provide at least the level of information required by the FDA’s new reporting requirement,” said Pushkin. Feinstein, DeLauro and others have previously introduced bills in Congress for a national breast density standard that ultimately stalled.
“This law is an important step forward, advanced by the efforts of many advocates, including those of the late Dr. Nancy Cappello of Are You Dense?. We are all grateful for the dedication and resolve of Sen. Dianne Feinstein and Rep. Rosa DeLauro who have championed patient notification in the several past congresses,” said Pushkin.
For more information: www.densebreast-info.org
Related Breast Density Content
Survey Shows Dense Breast Reporting Laws Increase Breast Density Awareness
Fake News: Having Dense Breast Tissue is No Big Deal
VIDEO: Breast Imaging and Risk Assessment at Northwestern Medicine Central DuPage


 April 18, 2024
April 18, 2024