March 26, 2018 — Auris Health Inc. announced U.S. Food and Drug Administration (FDA) clearance of the company’s Monarch Platform. The Monarch Platform integrates the latest advancements in robotics, software, data science and endoscope innovation, with the goal of improving patient outcomes, enhancing physician capabilities, and lowering costs to the healthcare system.
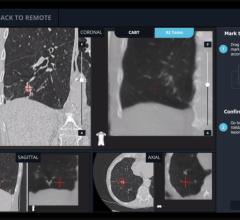
Auris’ first targeted disease state is lung cancer. The FDA cleared the platform for diagnostic and therapeutic bronchoscopic procedures. The goal of the technology is to enable more accurate diagnosis, and eventually treatment, of small and hard-to-reach nodules in the periphery of the lung.
Founded and led by surgical robotics pioneer Frederic Moll, M.D., Auris has raised more than $500 million in equity capital from leading technology investors including Mithril Capital Management, Lux Capital, Coatue Management and Highland Capital.
There are a variety of diagnostic options currently available for lung cancer, but all have limitations in accuracy, safety, or invasiveness. These limitations can lead to false positives, false negatives or side effects such as pneumothorax (collapsed lung) and hemorrhage, which may increase healthcare costs and extend hospital stays.
“Four hundred fifty people die every day in the United States due to lung cancer. It is the No. 1 cancer killer of both men and women in the world. Lung cancer screening has given us an opportunity to save some of these people by diagnosing the cancer early, while we have a chance to cure it. Despite the benefit, we still are limited by the current technology in making a diagnosis," said Michael Simoff, M.D., director of interventional pulmonology at Henry Ford Health System in Detroit. "The development of new advanced technology, like the Monarch Platform, could allow us the opportunity to make the diagnosis early, which translates directly to saving lives.”
The Monarch Platform is a flexible endoscopic technology that holds promise to fight lung cancer by allowing physicians to diagnose, and eventually treat, hard-to-reach, small peripheral nodules with greater precision. The platform utilizes a familiar controller-like interface that physicians use to navigate the flexible robotic endoscope to the periphery of the lung with improved reach, vision and control. Combining traditional endoscopic views into the lung with computer-assisted navigation based on 3-D models of the patient’s own lung anatomy, the Monarch Platform provides physicians with continuous bronchoscope vision throughout the entire procedure.
For more information: www.aurishealth.com


 March 21, 2024
March 21, 2024