April 26, 2016 — Philips announced that any hospital or healthcare facility with one of its indicated computed tomography (CT) models can now become a lung cancer screening center. Philips’ suite of CT solutions has achieved 510(k) clearance from the U.S. Food and Drug Administration (FDA) for low-dose lung cancer screening (LCS).
The suite includes 27 CT and positron emission tomography (PET)/CT models, as well as integrated software and services.
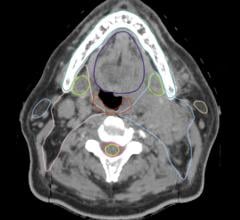
Lung cancer is the leading cause of cancer death among both men and women in the United States, and the source of one in four cancer deaths. Screenings are recommended by the Center for Medicare & Medicaid Services (CMS) to help identify the disease earlier in high-risk populations when treatment can be more successful. Early detection using this method has been shown to not only improve prognosis and treatment but decrease death rates up to 20 percent. In addition, if diagnosed in stage one, patients can have up to a 49 percent chance of surviving, compared to a diagnosis at stage three where the survival rate is as low as 5 percent.
“Lung cancer is the No. 1 cancer killer in America, taking more American lives than colon, breast and prostate cancer combined, and early detection is key to fighting this terrible disease,” said Brady J. McKee, M.D., radiologist at Lahey Hospital and Medical Center, which features the largest clinical lung cancer screening program in the country. “The work Philips has done to improve a provider’s screening capability not only benefits individual patients, but hospitals at each step of their Lung Cancer Screening program implementation and execution, which is often a very complicated process that includes community outreach, physician education, patient and data management, and reporting.”
Philips’ low-dose CT solutions now give healthcare organizations of varying clinical and economic needs — from community hospitals to multi-facility health systems — the ability to build robust lung cancer screening programs. Through its integrated radiology solutions and services, Philips can work with healthcare executives to better assess availability of existing scanners and to establish an enterprise-wide lung cancer program that will deliver advanced patient care and access.
Philips lung cancer screening solutions also provide several benefits for patients and referring physicians, including:
- Personalized management of radiation dose – The 27 models of Philips CT and PET/CT solutions qualified with the indications for use to perform CT lung cancer screening also utilize active dose management tools;
- Greater insights throughout the process – Referring physicians are better able to manage the process from end-to-end through “control center” software tools that enable digital access to patient tracking data that proves insight into scheduling, results and follow-up activities; and
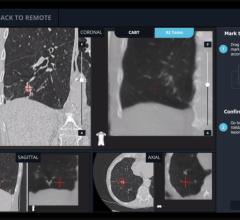
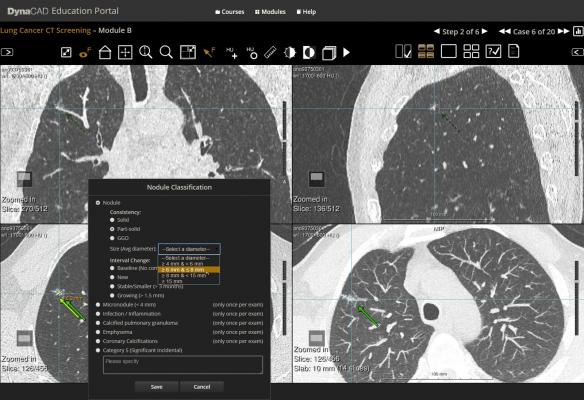
- Advanced image data sharing and analytics – Through its IntelliSpace Portal, Philips offers a comprehensive solution for detection, diagnostics and therapy follow-up. To address the increasing interest in pulmonary care, the latest version, IntelliSpace Portal 8.0, now includes the new CT Lung Nodule Assessment (LNA) application designed for a more efficient and longitudinal workflow to provide additional clinical decision support.
For more information: www.medical.philips.com

 April 16, 2024
April 16, 2024