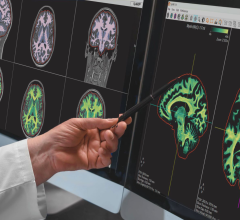
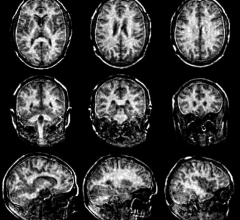
October 12, 2016 — The health tech company Icometrix obtained 510(k) clearance in September from the U.S. Food and Drug Administration (FDA) for their image quantification software, icobrain. The software quantifies key features of clinical magnetic resonance imaging (MRI) brain scans, which enable clinicians to monitor how their patient's brain changes over time.
As the only software currently available that has been scientifically validated and clinically approved for longitudinal measurements, according to the company, icobrain offers clinicians unrivalled access to unique information about disease progression and the effect of treatment upon brain structure.
"MRI biomarkers are becoming of vital importance in the clinical care path for various pathologies, including neurological disorders such as multiple sclerosis, traumatic brain injury and stroke. It is of paramount importance that the biomarker measurements are extremely reproducible and sensitive enough to detect relevant clinical changes", said Max Wintermark, M.D., professor of radiology and chief of neuroradiology at the Stanford University Medical Center. "The icometrix methods have been published in several scientific journals and validated by top centers worldwide, demonstrating the accuracy that is needed to translate measures from a research setting to routine clinical practice," Wintermark added.
Icometrix' analysis services help radiologists and physicians worldwide to improve and personalize the care of patients with a neurological disorder, such as multiple sclerosis, traumatic brain injury or dementia. Accessing the U.S. market is an important step and will lead to more evidence-based medicine by bringing automated MRI reports into clinical practice.
For more information: www.icometrix.com


 April 04, 2024
April 04, 2024