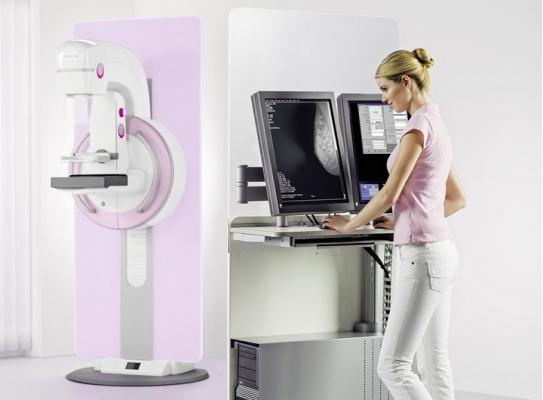
May 20, 2016 — Siemens Healthineers announced that the U.S. Food and Drug Administration (FDA) has approved the use of 3-D-only screening mammography utilizing the company’s Mammomat Inspiration with Tomosynthesis Option digital mammography system.
Siemens said the system is the first and only 3-D digital breast tomosynthesis (DBT) platform to be approved by the FDA as a stand-alone screening and diagnostic system; all other mammography systems on the market require a combination of 2-D and 3-D examinations.
FDA approval of the 3-D-only application follows a pivotal reader study in which participating radiologists demonstrated their ability to increase cancer detection at a lower radiation dose than combined 2-D and DBT. In the study, radiologists decreased average recall rates by an average of 19 percent without the need for a 2-D image.
Siemens’ Tomosynthesis Only Option is available on the company’s Mammomat Inspiration and Mammomat Inspiration Prime Edition digital mammography systems.
For more information: www.healthcare.siemens.com


 April 12, 2024
April 12, 2024