If you enjoy this content, please share it with a colleague
RELATED CONTENT
Technology
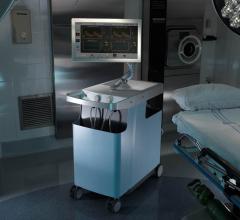
PhysioSonics announced the U.S. Food and Drug Administration (FDA) has granted the Presto 1000 Flow Monitor system 510(k) clearance for cranial blood flow monitoring. In addition, the company announced completion of Series B add-on debt financing, closing with $1.05 million.

 February 01, 2013
February 01, 2013 