If you enjoy this content, please share it with a colleague
ImaginAb Inc. U.S.
RELATED CONTENT
ImaginAb Inc. announced the enrollment of the first patient in the Phase II clinical trial of the company’s CD8+ T Cell imaging agent. The trial will enroll metastatic cancer patients and will study the correlation of imaging signals observed using ImaginAb’s CD8+ T cell ImmunoPET imaging agent, standard-of-care scans and immunohistochemistry analysis of CD8 in biopsied tissues.
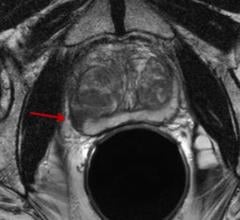
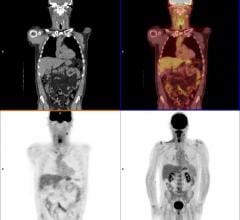
ImaginAb Inc. has released interim results from a Phase 2 clinical trial of IAB2M, the company's proprietary imaging agent for management of prostate cancer. IAB2M demonstrated superior performance in detecting disease compared with ProstaScint and conventional imaging technologies including computed tomography (CT) scans, magnetic resonance imaging (MRI) scans and bone scans. In addition, the agent accurately detected metastatic disease in normal-sized lymph nodes, overcoming a major limitation of existing technologies and addressing a key clinical need.
June 9, 2014 — ImaginAb Inc., in collaboration with Memorial Sloan-Kettering Cancer Center, will present at the 2014 annual meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI) initial clinical data with an anti-PSMA (prostate-specific membrane antigen) recombinant antibody fragment (a "minibody") derived from the huJ591 monoclonal antibody, in patients with metastatic prostate cancer.

 January 30, 2019
January 30, 2019